The incidence of dyslipidemia is significantly higher in patients treated with antiretroviral regimens containing ritonavir compared to other regimens not containing ritonavir.1 Moreover, protease inhibitors (PIs) are not equally prone to impair the lipid profile. Regimens with lopinavir/ritonavir (LPV/r) and fosamprenavir/ritonavir (FPV/r) indeed, induce greater elevations in lipid parameters than saquinavir/ritonavir (SQV/r) or atazanavir/ritonavir (ATV/r) or darunavir/ritonavir (DRV/r).2 The objective of this study was to explore the impact on the lipid profile of switching from “older PIs” to DRV/r in virologically suppressed HIV infected patients after 144 weeks of observation. A prospective open label single center study enrolled a total of 13 Caucasian subjects who switched to DRV/r at dosage 800mg/100mg, without keeping the same NRTI backbone.
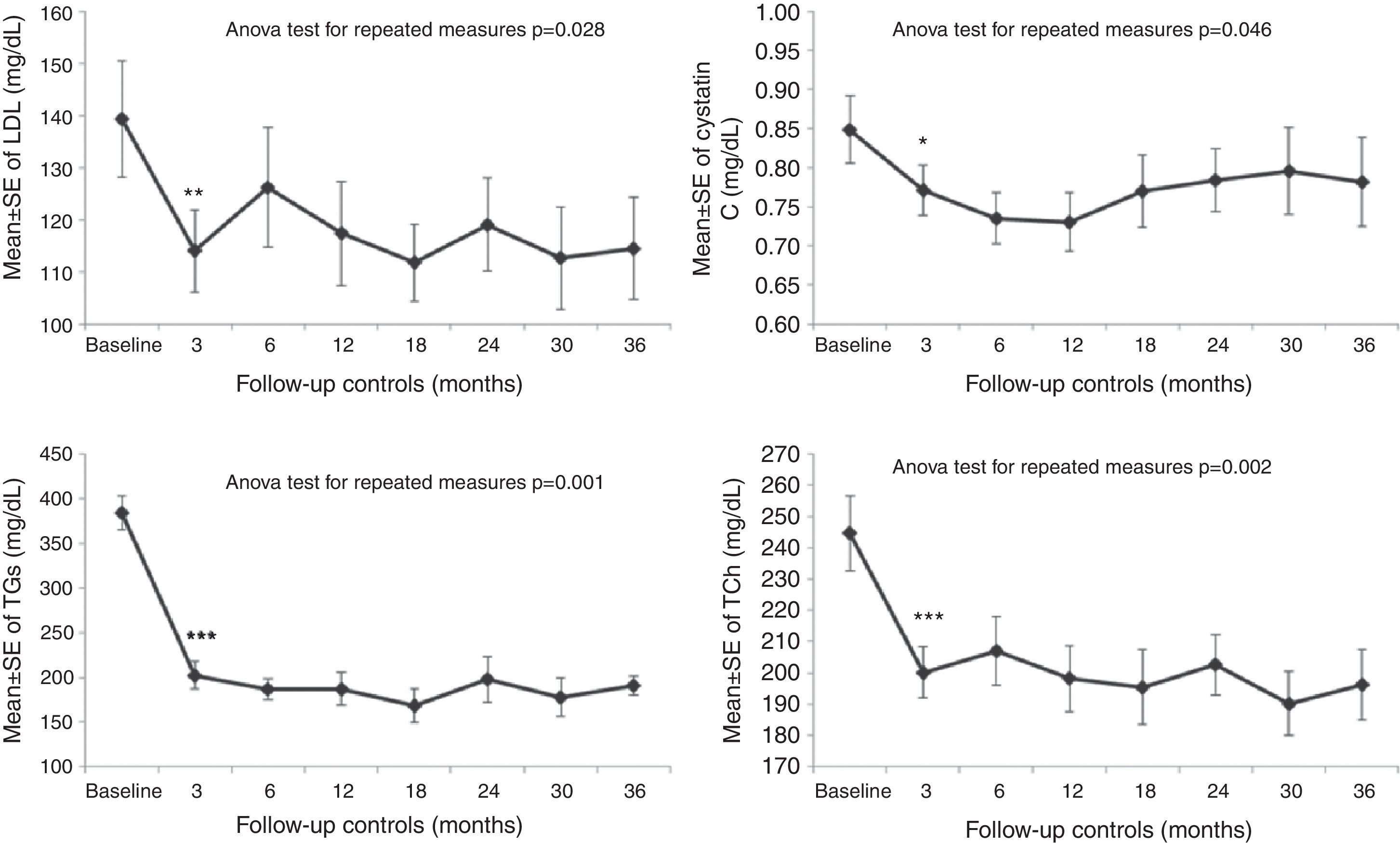
The switch to DRV/r reduced triglycerides levels, total and LDL cholesterol right after three months. Similar improvements remained stable during the follow up period. At the end of the study a significant reduction of triglycerides (TGs), total (TCh) and LDL cholesterol were in the order of 47%, 18%, and 18%, respectively (TGs at T0 383.7±70.2 versus T12 202.4±60.7mg/dL, p<0.001, TCh at T0 244.7±51.3 versus T12 200.1±31.8mg/dL, p<0.001; LDL at T0 139.3±43.4, versus T12 114.1±30.7mg/dL, p<0.01) (Fig. 1). In addition, the TCh/HDL ratio has also improved (p=0.004). Increases in CD4+ cell counts and in CD4/CD8 ratio (p<0.05) were also observed. Serum cystatin C values showed a statistically significant decrease during the follow up period (T0 0.84±0.16 versus 0.78±0.20mg/dL, p=0.046). After 144 weeks the switch from LPV/r or FPV/r to DRV/r in HIV infected patients with triglycerides higher than 200mg/dL, induced a sustained improvement on the lipid parameters, and specifically a reduction of main lipid makers. These effects may be explained by the reduced booster dose of ritonavir (200mg to 100mg), and by a smaller impact of DRV on lipid parameters as compared to other PIs containing regimens, as previously showed. In this study, it should be pointed out that this improvement was long-lasting with excellent HIV-RNA control; all patients completed the study without virological failure. Of note, we observed an improvement in TCh/HDL ratio another strong surrogate marker for predicting cardiovascular events as previously demonstrated in HIV negative population.3 In this study we showed an improvement in different surrogate markers of CD risk, such as TGs, TCh, LDL, TCh/HDL ratio, confirming data previously published4 and the long-term efficacy of this strategy. An interesting result of this study is the significant reduction of cystatin C serum levels at every time point of observation reaching 8% at 144 weeks. Moreover, a significant decrease of microalbuminuria was observed with a reduction of 35% at the end of the study, in patients without renal impairment or decreased glomerular filtration rate. This is the first significant data on microalbuminuria after switching PIs. Cardiovascular risk reduction is also supported by improvement in cystatin C and microalbuminuria which are important markers of inflammatory status, CD risk, and mortality.5 The continuous increase in CD4+ cell counts after switching PIs and a remarkable significant improvement in CD4/CD8 ratio are surrogate markers of important immune reconstitution. In conclusion, in HIV infected patients on antiretroviral therapy and with abnormal lipid profiles, switching to DRV/r must be considered for long term containment of the metabolic abnormalities.
The authors declare no conflicts of interest.