Since antimicrobial resistance among uropathogens against current first line agents has affected the management of severe urinary tract infection, we determined the likelihood that antibiotic regimens achieve bactericidal pharmacodynamic exposures using Monte Carlo simulation for five antimicrobials (ciprofloxacin, ceftriaxone, piperacillin/tazobactam, ertapenem, and meropenem) commonly prescribed as initial empirical treatment of inpatients with severe community acquired urinary tract infections. Minimum inhibitory concentration determination by Etest was performed for 205 Brazilian community urinary tract infection Escherichia coli strains from 2008 to 2012 and 74 E. coli bloodstream strains recovered from a surveillance study. Pharmacodynamic exposure was modeled via a 5000 subject Monte Carlo simulation. All isolates were susceptible to ertapenem and meropenem. Piperacillin/tazobactam, ceftriaxone and ciprofloxacin showed 100%, 97.5% and 83.3% susceptibility among outpatient isolates and 98.6%, 75.7% and 64.3% among inpatient isolates, respectively. Against outpatient isolates, all drugs except ciprofloxacin (82.7% in aggressive and 77.6% in conservative scenarios) achieved high cumulative fraction of response: carbapenems and piperacillin/tazobactam cumulative fraction of responses were close to 100%, and ceftriaxone cumulative fraction of response was 97.5%. Similar results were observed against inpatients isolates for carbapenems (100%) and piperacillin/tazobactam (98.4%), whereas ceftriaxone achieved only 76.9% bactericidal cumulative fraction of response and ciprofloxacin 61.9% (aggressive scenario) and 56.7% (conservative scenario) respectively. Based on this model, standard doses of beta-lactams were predicted to deliver sufficient pharmacodynamic exposure for outpatients. However, ceftriaxone should be avoided for inpatients and ciprofloxacin empirical prescription should be avoided in both inpatients and outpatients with complicated urinary tract infection.
Urinary tract infection (UTI) is the most common bacterial infection and acute pyelonephritis is a severe form of UTI with symptoms that range from mild discomfort to life threatening illness or death.1,2 The incidence of pyelonephritis requiring hospitalization is 3–4 cases per 10,000 population among women and 1–2 cases per 10,000 population among men, with an estimation of direct and indirect costs of US$ 2.1 billion and in hospital mortality of 7.3 per 1000 hospitalizations for females and 16.5 per 1000 for males.2–4 The bacteriology of pyelonephritis is similar to the other UTIs, the majority of them being caused by Escherichia coli.3,5
The management of UTIs is usually empirical, based on the predictable spectrum of etiologic agents and their susceptibility patterns. Due to the emergence of antimicrobial resistance among uropathogens, the safety and effectiveness of empirical therapy have been affected.6 Recently, ex vivo pharmacodynamic (Pd) models (based on Monte Carlo simulation) have been used to help determine the best regimens or dosages for a specific patient population, by integrating an agent's in vitro potency distribution (i.e., minimum inhibitory concentrations [MICs]) with the pharmacokinetic (Pk) profile of a drug.7
Regional surveillance studies show that antimicrobial resistance against currently prescribed first line agents (trimethoprim/sulfamethoxazole and ciprofloxacin) for treatment of community acquired UTIs in Brazil is increasing.8,9 Since the resistance patterns of E. coli strains causing UTI vary considerably between regions and countries, specific treatment regimens based on local susceptibility patterns are necessary.5,9 The aim of this study was to compare the pharmocodynamic profiles of five antimicrobials (ciprofloxacin, ceftriaxone, piperacillin/tazobactam, ertapenem and meropenem) commonly prescribed as initial empirical therapy in hospitals for the treatment of inpatients with community acquired UTIs, usually complicated or pyelonephritis.
Materials and methodsMicrobiologyBacterial isolatesThe present study included a total of 279 bacterial isolates; 100 isolates of outpatients stored at the culture collection of the Special Laboratory of Clinical Microbiology (LEMC) since 2008, 105 isolates of outpatients prospectively collected between August and November 2012, and 74 nosocomial isolates from the first bacteremia at 16 hospitals in Brazil participants of the BrSCOPE study.10 All isolates were identified with the BD Phoenix 5.1TM (Becton Dickinson, Sparks, MD 21152, USA) and/or biochemical identification methods. Only one isolate per patient was included in the study.
Minimal inhibitory concentration (MIC) testingThe MIC was determined by epsilometric test (Etest®, BioMérieux, Marcy l’Etoile, France) for ertapenem (0.002–32), meropenem (0.002–32), ciprofloxacin (0.002–32), ceftriaxone (0.016–256) and piperacillin/tazobactam (0.016–256), following the recommendations of the manufacturer. Quality control was performed by testing E. coli ATCC 25,922® and P. aeruginosa ATCC® 27,853, with all results within expected ranges.
Samples were subcultured onto blood agar and then onto MacConkey agar to ensure viability and purity. Individual colonies were picked from 18 to 24h plates and suspended in 0.85% saline to a turbidity of a 0.5 McFarland standard. Cotton swabs were used to transfer the inoculum to the plates with Mueller Hinton agar (Oxoid, Basingstoke, Inglaterra), which were dried before the Etest® strips were applied. For each strain, the five antibiotics were arranged in a single plate. Plates were incubated for 18–24h at 35°C. Readings and interpretations were performed according to National Committee for Clinical Laboratory Standards (NCCLS).11
Pk and Pd dataPk dataPk data were obtained from previously published studies in healthy volunteers.12–14 For studies to be considered, they had to be conducted in at least 10 healthy volunteers, describing the assay used to determine drug concentrations, and present mean and standard deviation results for the total body clearance in liters per hour (CL), volume of distribution of the central compartment (Vc) and other pertinent Pk parameters. Because published reports with the above characteristics were not identified for piperacillin/tazobactam and ceftriaxone, Pk data for these drugs were taken from published studies15,16 in critical patients (no studies describing the covariance matrix in healthy volunteers). The Pk data was then used in the Monte Carlo simulation models. A 5000 patient Monte Carlo simulation (Oracle Crystal Ball Fusion Edition, 2010) was conducted to calculate estimates of the amount of time in which the free or non-protein-bound drug concentration exceeds the MIC of the organism (fT>MIC) or the AUC/MIC ratio for each antibiotic regimen/bacterial population combination. The simulations were conducted as previously published.17,18 Drug dosages chosen were based on the following commonly prescribed regimens, administered as 30-min intravenous (IV) infusions: ertapenem 1g every 24h (q24h), meropenem 1g every 8h (q8h), piperacillin/tazobactam 4.5g every 8h (q8h), ceftriaxone 1g every 12h (q12h) and ciprofloxacin 400mg every 12h (q12h). Pd exposures, as measured by fT>MIC, were simulated for all beta-lactam regimens against all pathogen groups. A two compartment IV bolus equation was used to calculate % fT>MIC for the beta lactams. For ciprofloxacin, the total drug AUC was calculated as the daily dose divided by the total body clearance (ClT).
Pd modelValues for fT>MIC and AUC/MIC were plotted on frequency curves for further analysis. The probabilities of obtaining a fT>MIC of 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, and 100% were calculated for all the beta-lactams at increasing MICs in doubling dilutions. Pd targets of 40% fT>MIC and 50% fT>MIC were considered bactericidal for carbapenems and for ceftriaxone and piperacillin/tazobactam, respectively. We also calculated the cumulative fT>MIC of beta-lactams, previously proposed as Pd threshold for UTI. Microbiological cure were achieved attaining a cumulative fT>MIC above 30h.19 The probability for achieving a total AUC/MIC ratio greater than or equal to 62.5, 75, 100, 125, and 150 were calculated for ciprofloxacin. The Pd target of AUC/MIC at least 62.5 (conservative scenario, half of the target for bloodstream infection) and 125 (aggressive scenario, similar to the target for bloodstream infection) were chosen as a Pd breakpoint for ciprofloxacin. These probabilities of target attainment were applied to the MIC distributions for the above pathogens to calculate the cumulative fraction of response (CFR).
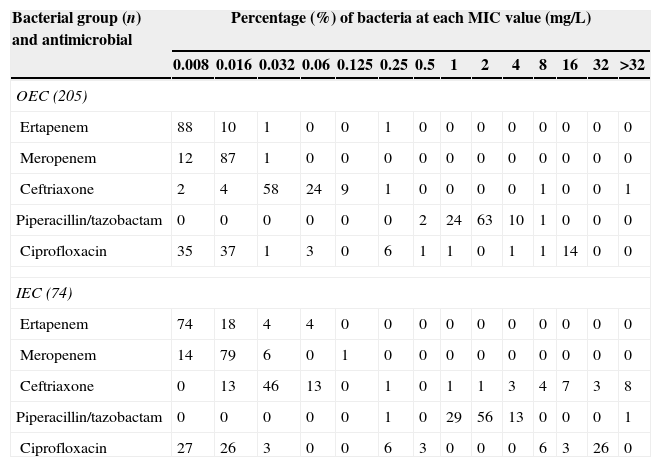
ResultsIn vitro activityTable 1 summarizes the results of the in vitro susceptibility testing. The resistance profile of outpatient isolates from 2008 to 2012 was similar. All isolates were susceptible to ertapenem and meropenem (MIC range 0.008–0.25μg/mL and MIC range 0.008–0.032μg/mL, respectively); 100% of outpatient isolates (MIC range 0.5–8μg/mL) and 98.6% of inpatient isolates (MIC range 0.25–256μg/mL) showed susceptibility to piperacillin/tazobactam. Higher resistance rates against ceftriaxone were observed for inpatient E. coli (75.7% susceptibility, MIC range 0.016–256μg/mL) when compared to outpatient E. coli (97.5% susceptibility, MIC range 0.008–256μg/mL). Among the antibiotics tested, ciprofloxacin had the lowest susceptibility rates against the outpatient E. coli (83.3%, MIC range 0.008–0.032μg/mL) and inpatient E. coli (64.3%, MIC range 0.008–32μg/mL).
MIC distribution for outpatient E. coli (OEC) and inpatient E. coli (IEC) included in this study.
| Bacterial group (n) and antimicrobial | Percentage (%) of bacteria at each MIC value (mg/L) | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0.008 | 0.016 | 0.032 | 0.06 | 0.125 | 0.25 | 0.5 | 1 | 2 | 4 | 8 | 16 | 32 | >32 | |
| OEC (205) | ||||||||||||||
| Ertapenem | 88 | 10 | 1 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Meropenem | 12 | 87 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Ceftriaxone | 2 | 4 | 58 | 24 | 9 | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 1 |
| Piperacillin/tazobactam | 0 | 0 | 0 | 0 | 0 | 0 | 2 | 24 | 63 | 10 | 1 | 0 | 0 | 0 |
| Ciprofloxacin | 35 | 37 | 1 | 3 | 0 | 6 | 1 | 1 | 0 | 1 | 1 | 14 | 0 | 0 |
| IEC (74) | ||||||||||||||
| Ertapenem | 74 | 18 | 4 | 4 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Meropenem | 14 | 79 | 6 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Ceftriaxone | 0 | 13 | 46 | 13 | 0 | 1 | 0 | 1 | 1 | 3 | 4 | 7 | 3 | 8 |
| Piperacillin/tazobactam | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 29 | 56 | 13 | 0 | 0 | 0 | 1 |
| Ciprofloxacin | 27 | 26 | 3 | 0 | 0 | 6 | 3 | 0 | 0 | 0 | 6 | 3 | 26 | 0 |
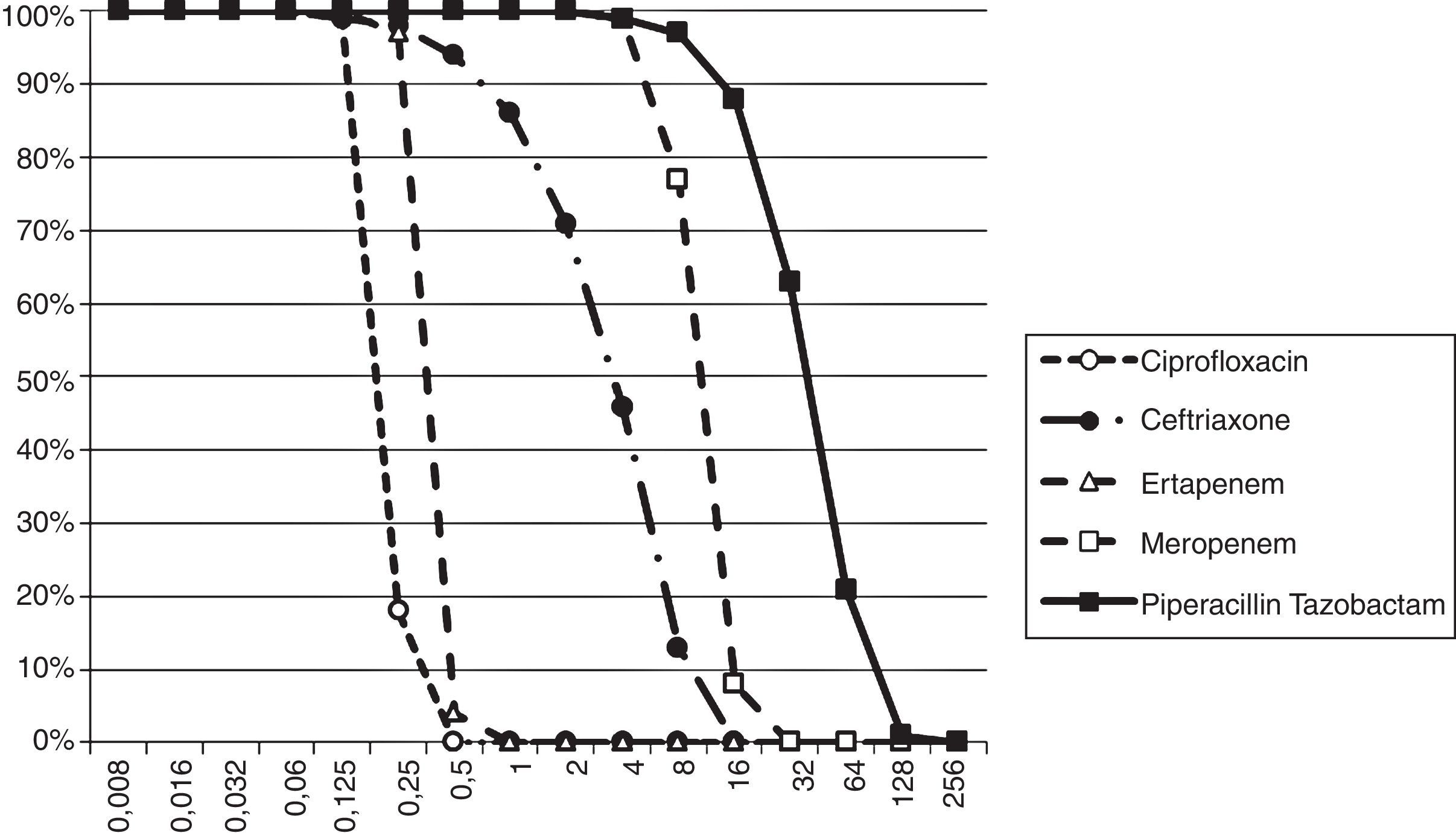
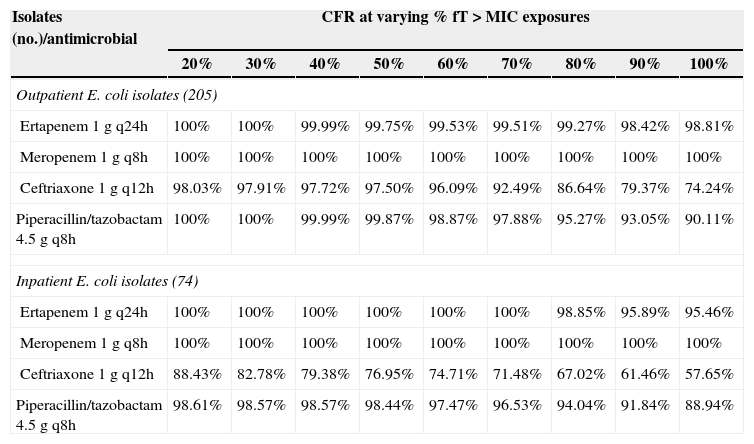
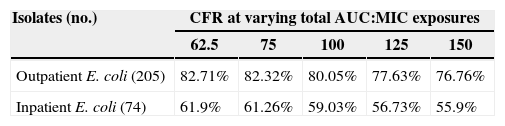
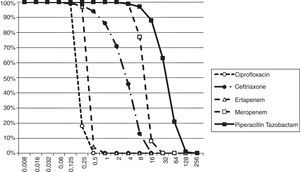
The Probability of Target Attainment (PTA) for beta-lactams and ciprofloxacin over a range of MICs is indicated in Fig. 1. The CFRs for each beta-lactam at varying targets for % fT>MIC are listed in Table 2 and the CFR for ciprofloxacin at AUC/MIC target of 62.5–150 is presented in Table 3. Carbapenems demonstrated high likelihood of achieving the bactericidal breakpoint of 40% fT>MIC and piperacillin/tazobactam at 50% fT>MIC in both scenarios. In contrast, ceftriaxone achieved a CFR superior to 95% only against outpatient isolates. Ciprofloxacin had poor CFR against outpatient (82.7%, 77.6%) and inpatient (61.9%, 56.7%) isolates at the target exposure of total AUC/MIC>62.5 and AUC/MIC>125, respectively.
Cumulative fraction of response (CFR) at varying % fT>MIC for beta-lactams against outpatient E. coli and inpatient E. coli isolates from Brazil.
| Isolates (no.)/antimicrobial | CFR at varying % fT>MIC exposures | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 20% | 30% | 40% | 50% | 60% | 70% | 80% | 90% | 100% | |
| Outpatient E. coli isolates (205) | |||||||||
| Ertapenem 1g q24h | 100% | 100% | 99.99% | 99.75% | 99.53% | 99.51% | 99.27% | 98.42% | 98.81% |
| Meropenem 1g q8h | 100% | 100% | 100% | 100% | 100% | 100% | 100% | 100% | 100% |
| Ceftriaxone 1g q12h | 98.03% | 97.91% | 97.72% | 97.50% | 96.09% | 92.49% | 86.64% | 79.37% | 74.24% |
| Piperacillin/tazobactam 4.5g q8h | 100% | 100% | 99.99% | 99.87% | 98.87% | 97.88% | 95.27% | 93.05% | 90.11% |
| Inpatient E. coli isolates (74) | |||||||||
| Ertapenem 1g q24h | 100% | 100% | 100% | 100% | 100% | 100% | 98.85% | 95.89% | 95.46% |
| Meropenem 1g q8h | 100% | 100% | 100% | 100% | 100% | 100% | 100% | 100% | 100% |
| Ceftriaxone 1g q12h | 88.43% | 82.78% | 79.38% | 76.95% | 74.71% | 71.48% | 67.02% | 61.46% | 57.65% |
| Piperacillin/tazobactam 4.5g q8h | 98.61% | 98.57% | 98.57% | 98.44% | 97.47% | 96.53% | 94.04% | 91.84% | 88.94% |
Cumulative fraction of response (CFR) for varying total AUC:MIC for ciprofloxacin 400mg q12h against outpatient E. coli and inpatient E. coli isolates from Brazil.
| Isolates (no.) | CFR at varying total AUC:MIC exposures | ||||
|---|---|---|---|---|---|
| 62.5 | 75 | 100 | 125 | 150 | |
| Outpatient E. coli (205) | 82.71% | 82.32% | 80.05% | 77.63% | 76.76% |
| Inpatient E. coli (74) | 61.9% | 61.26% | 59.03% | 56.73% | 55.9% |
Considering a treatment duration of seven days, the cumulative fT>MIC superior to 30h was achieved at MIC concentration of 8μg/mL for ceftriaxone, 8μg/mL for meropenem, 1μg/mL for ertapenem and 64μg/mL for piperacillin/tazobactam.
DiscussionIncreasing rates of resistance to antimicrobials commonly used for treating UTIs caused by E. coli among outpatients has been reported in recent years.20 In this context, this study compared the antimicrobial susceptibility of community-acquired UTI E. coli strains between two periods, since local susceptibility patterns are essential for appropriate antimicrobial prescribing. The study also included nosocomial isolates from the first bacteremia episode. Despite high variability in the origin of bacteremia reported in the literature, the urinary tract is one of the most frequent sources for E. coli bacteremia.21 For this reason, the authors considered the inclusion of these isolates in the study as relevant.
The sensitivity profile of outpatient E. coli was similar between 2008 and 2012. For ciprofloxacin, local surveillance studies on uropathogens in community-acquired infections9,22 also found decreased susceptibility pattern of outpatient E. coli, especially since the year 2000. Our present results are similar to those described locally by Rocha et al.,22 which obtained 82.2% in isolates of 2009. For other antimicrobials tested, our study showed results similar to the SENTRY study: low prevalence of ceftriaxone and piperacillin/tazobactam resistance and no resistance against carbapenems.9
The Monte Carlo Simulation has already been used to forecast the efficacy of different therapeutic regimens in multiple scenarios, such as bloodstream infections, community-acquired pneumonia and ventilator-associated pneumonia.17,23,24 To the best of our knowledge, our study is the first report to apply the Monte Carlo Simulation evaluating the likelihood of achieving bactericidal Pd targets in UTIs.
As for the Pd targets adopted, few studies have established specific Pd thresholds for UTI. Deguchi et al.,25 determined the Pd target of levofloxacin for microbiological cure in patients with complicated UTI based on the plasma concentration of the drug and the threshold of AUC/MIC for predicting microbiological cure was lower than those in other kind of infections.
No studies correlating a Pd target of ciprofloxacin plasma concentration and microbiological cure for UTI were found in our review. Since ciprofloxacin and levofloxacin are fluoroquinolones and targets within antibiotic classes tend to be similar, we proposed the two scenarios previously described: an aggressive and a conservative one for ciprofloxacin.
With respect to beta-lactam antibiotics, a cumulative fT>MIC around 30h was correlated to maximum cure rate for serum aminopenicillin concentration.19 Unfortunately, no study correlating microbiological cure for UTI and fT>MIC ratio for meropenem, ertapenem, piperacilin/tazobactam and ceftriaxone were found. Considering the target for aminopenicillin similar to the drugs included in this study, those results might not be applicable on real life scenario since an 8μg/mL MIC for meropenem and a 64μg/mL MIC for piperacillin/tazobactam would be considered intermediate based on CLSI breakpoints, and a 16μg/mL MIC for ceftriaxone, resistant. For renal tissue infection, the serum or plasma concentration is a useful marker for the antibiotic concentrations in the renal tissue.19 As Pd targets for beta-lactams in severe UTI are not well elucidated, for the present simulation we considered the established targets for bloodstream infections.
Initially, we estimated the variability for the Pk parameters derived from healthy volunteers. Pk data from critical patients were chosen for piperacillin/tazobactam and ceftriaxone due to a lack of published reports which met the inclusion criteria for our study. DeRyke et al.,18 found CFR largely similar for piperacillin/tazobactam whether Pk of patients or those of healthy volunteers were used. Concerning ceftriaxone, in spite of a weak increase of Vc and wide inter-patient variability observed in critical patients, total serum concentrations were similar to that observed in normal patients.26
Current recommendation advises not to prescribe ciprofloxacin as empirical treatment for UTI if local resistance exceeds 10%.5 Surveillance studies had already demonstrated that ciprofloxacin resistance in Brazil overcomes such percentage.18,19 Our study corroborates epidemiological findings, which impacts on a low probability of achieving a bactericidal exposure (CFR of 77.6% for outpatients and 56.7% for inpatients considering an AUC/MIC target of at least 125). Even if lower Pd targets were chosen, CFR stayed consistent (≤5% decreases).
Based on the simulated PTAs ciprofloxacin should be prescribed only when MIC was equal or below 0.25μg/mL, despite the susceptibility threshold currently considered. Inconsistency between susceptibility and the predicted efficacy of antimicrobials has already been reported since CLSI-defined breakpoints for Gram negative bacilli might overestimate susceptibility in two to fourfold greater than those estimated by Pd simulations.27
Ceftriaxone is a good option to treat severe UTI infection in outpatients based on our simulations. Although dissemination of clones producing ESBL of community-acquired E. coli infections were already reported intercontinentally,8 almost all outpatient isolates were susceptible to ceftriaxone. We observed a high percentage of non-susceptibility against ceftriaxone (25.7%) in inpatient isolates, which decreases the probability of achieving bactericidal exposures (76.9%) with this regimen.
In this analysis, piperacillin/tazobactam, ertapenem and meropenem achieved the highest CFRs in both scenarios. Previous Pk/Pd studies also found a high likelihood of achieving optimal Pd target exposure for meropenem against E. coli isolates.17,18,27
Similar to the derived result from Pd simulation of ciprofloxacin, ertapenem presented a fourfold discrepancy between CLSI defined breakpoint and those derived from stochastic determination. Although ertapenem had similar in vitro activity to meropenem, its ability to achieve high bactericidal Pd exposure will depend on the presence of a highly susceptible population,17 which was reported in this study.
In spite of high CFR observed in present study, lower CFRs of piperacillin/tazobactam against E. coli isolates was previously demonstrated.24 Beta-lactam/beta-lactam inhibitor combination is an effective option to treat ESBL – E. coli bloodstream infection originating from the urinary tract and should be considered a reasonable alternative to carbapenems if active in vitro.21
Certain assumptions are made for analyses performed with Monte Carlo simulations. First, although the resistance rates in our study were similar to previous surveillance studies, it may not reflect isolate frequency and/or susceptibilities at all institutions. Second, we used Pk models derived from healthy and infected adult patients, most of whom had normal renal function. Dose adjustments might be necessary to attain similar results for patients with renal insufficiency or pediatric population. Finally, further studies are required to elucidate the Pd threshold for ciprofloxacin and beta-lactams necessary to provide microbiological cure in UTIs.
On the basis of this model using microbiology data from outpatient and inpatient urinary isolates, standard doses of beta-lactams were predicted to deliver sufficient Pd exposure for outpatients. In contrast, the low probability predicted for ceftriaxone suggests that this drug should not be prescribed in patients who have history of recent hospitalization, early onset of in-hospital E. coli bacteremia or are in close relationship with healthcare units. Ciprofloxacin prescription should be discouraged even in community-acquired severe ITU.
FundingThis study was supported by Merck Sharp & Dohme. The pharmaceutical company was neither involved nor had any interference with data collection, interpretation of findings or preparation of the manuscript.
Ethical approvalThis study was approved by the Ethics Committee of Universidade Federal de São Paulo (UNIFESP).
Conflicts of interestThe authors declare no conflicts of interest.
This study was partially presented as an abstract at the 23rd European Congress of Clinical Microbiology and Infectious Diseases, 2013 (abstract 2872).