The identification of the best strategy to manage cytomegalovirus infection is hampered by uncertainties regarding the risk/benefit ratios of universal prophylaxis versus preemptive therapy, the impact of indirect cytomegalovirus effects and the associated costs. This study investigated the efficacy and safety of targeted preemptive therapy according to perceived risk of cytomegalovirus infection after kidney transplantation.
Methods144 adult kidney transplant recipients were enrolled in this 12-month study. None received cytomegalovirus pharmacological prophylaxis. Only high risk patients (positive donor/negative recipient (D+/R−), use of induction therapy with antithymocyte globulin, treatment of rejection) received preemptive therapy based on the result of pp65 antigenemia test. Low-risk patients with symptoms related to cytomegalovirus were screened for pp65 antigenemia and treatment initiated if confirmed cytomegalovirus disease. Blinded cytomegalovirus DNAemia was collected weekly during the first three months.
ResultsThe incidence of cytomegalovirus infection was 34% and cytomegalovirus disease was 17%. The incidence was 25% in D+/R−, 69% in those receiving induction with rabbit antithymocite globulin (r-ATG), 46% in those treated for acute rejection, and 28% in low risk patients. By week 3 DNAemia was observed in 30% of patients who were not treated for cytomegalovirus infection/disease, and values ≥2.169UI/mL showed 61% sensitivity and 85% specificity to detect cytomegalovirus disease (AUC=0.849±0.042, p<0.001). Using multivariate analysis, only anti-thymocyte globulin induction was associated with cytomegalovirus infection/disease whereas only expanded donor criteria and renal function at 30 days were associated with renal function 12 months after transplantation.
ConclusionTargeted preemptive therapy in patients with perceived higher risk for cytomegalovirus infection/disease was effective in preventing severe clinical presentation, including tissue invasive and late cytomegalovirus infection. This strategy is associated with direct and indirect cost-savings.
Cytomegalovirus (CMV) infection remains one of the most common complications affecting transplant recipients,1–3 and has been associated with a wide spectrum of adverse events, including impaired graft function and occasional mortality.2 Strategies for the prevention of CMV infection have been associated with significant reduction in incidence and severity of disease.4,5 Despite the large variability in the management of CMV infection among transplant centers two main strategies, namely universal prophylaxis or preemptive therapy, are currently used. Moreover, each of these strategies has significant variability in clinical practice, including the type of cell or molecular diagnosis, antiviral therapies, frequency of monitoring, and criteria for initiating and stopping treatment.6
The incidence of CMV infection is primarily associated with pretransplant serological status (donor positive, recipient negative, D+/R−), use of induction with rabbit antithymocyte globulin (r-ATG), maintenance immunosuppressive regimen, and treatment for acute rejection.7–9 Although the use of universal prophylaxis has increased since the availability of valganciclovir, there is still a debate related to the superiority of this strategy over the preemptive approach.10–12 Data from prospective randomized trials have shown superiority of prophylaxis over preemptive therapy only in the D+/R− high risk population. Moreover, the associated costs may be a decisive factor in several countries with no pre-specified reimbursement.13
One of the major concerns regarding preemptive therapy is that it may not prevent the indirect effects of CMV infection, despite conflicting data on its association with graft and patient survival.14–16 In addition, second episodes of CMV viral replication have been reported in up to 30% of patients, some of which require further therapeutic intervention.17,18 On the other hand, in one recent study 53% of kidney transplant recipients receiving tacrolimus and mycophenolate presented transient CMV viremia that was cleared spontaneously without anti-viral treatment.19
Considering these uncertainties, this study investigated the use of targeted preemptive therapy for CMV infection in a cohort of kidney transplant patients receiving different immunosuppressive regimens and no pharmacological CMV infection prophylaxis.
MethodsStudy populationThis was a single center, prospective 12-month study in de novo kidney transplant recipients. The study was conducted in compliance with Good Clinical Practice guidelines, in accordance with the Declaration of Helsinki and was approved by the local Ethics Committee. We screened patients older than 18 years, recipients of first or second kidney only transplants from brain-dead deceased or living-related donors. All patients signed an informed consent form.
ImmunosuppressionPatients who received kidneys from expanded criteria donors20 received induction therapy with 4×1.5mg/kg doses of r-ATG every other day from day 1 after transplantation. The maintenance therapy consisted of 1440mg BID of mycophenolate sodium. Tacrolimus (TAC) at initial dose of 0.05mg/kg BID was associated from day 7. TAC doses were adjusted to maintain whole blood trough concentrations between 6 and 8ng/mL during the first three months and between 4 and 6ng/mL thereafter.
Patients who received kidneys from standard deceased or living donors received no induction therapy. The maintenance therapy consisted of initial TAC doses of 0.1–0.15mg/kg BID to maintain whole blood trough concentrations between 10 and 15ng/mL during the first month and between 8 and 12ng/mL thereafter. Patients also received 1440mg BID of mycophenolate sodium or azathioprine at daily doses of 2mg/kg. All patients received 1g methylprednisolone prior to graft revascularization and an initial 0.5mg/kg of prednisone from day 1 tapered to 5mg by day 45.
ProphylaxisNo pharmacological prophylaxis for CMV infection was used. All patients received oral trimethoprim-sulfamethoxazole for at least six months as prophylaxis for Pneumocystis jiroveci pneumonia or urinary tract infection, as per local practice. All patients received albendazole 400mg for 5 days as prophylaxis for parasitic infections.
CMV management strategyPer local practice, only patients deemed high risk for CMV infection were monitored for CMV viral replication. High risk patients were D+/R−, those who received induction therapy with r-ATG, and those treated for acute rejection. These patients were monitored every other week beginning on day 21 post transplant for CMV infection using the pp65 antigenemia test. CMV infection in asymptomatic patients was defined as the presence of >10 pp65 positive cells/200,000 leucocytes. CMV disease in symptomatic patients (fever, malaise, leukopenia, and/or thrombocytopenia) was defined as the presence of any pp65 positive cells/200,000 leucocytes. CMV tissue invasive disease was confirmed by direct identification of the virus in any tissue. Treatment of CMV infection or disease consisted of intravenous 5mg/kg BID ganciclovir, adjusted for renal function. Treatment was monitored weekly with pp65 antigenemia test and extended for one additional week after a negative antigenemia test.
Recurrence was defined as the need for a new treatment after complete remission of the last episode.
Quantitative nucleic acid amplification test (QNAT)Patients were monitored for viral replication every week for three months. Investigators were blinded to QNAT results. CMV DNAemia was quantified in EDTA human plasma samples using the COBAS® AmpliPrep/COBAS® TaqMan® CMV test (Roche Molecular Diagnostics) according to manufacturer instructions. The test allowed for determination of CMV DNA ranging from 150 to 10,000,000copies/mL. A copy of CMV DNA is equivalent to 0.91 International Units (IU) based on the 1st WHO International Standard for human CMV techniques for nucleic acid amplification.21
Efficacy and safetyDelayed graft function was defined as the need for at least one dialysis during the first week after transplantation. All patients with suspected acute rejection underwent a core graft biopsy and were classified according to Banff 2007 criteria.22 Treatment with methylprednisolone or r-ATG was considered according to clinical and histological severity analysis. All corticosteroid-resistant acute rejection episodes were confirmed by biopsy and treated with r-ATG. Glomerular filtration rate (GFR) was calculated using the MDRD formula23 at 1 and 12 months.
Statistical analysisDescriptive analyses were summarized as mean and standard deviation or proportions. Categorical variables were compared using Chi-square test. Continuous variables were compared using ANOVA test. Univariate and multivariate logistic regression analyses were performed to identify risk factors associated with CMV infection/disease and with renal function at 12 months. All statistical analyses were performed using the SPSS 18 standard software (SPSS Inc., Chicago, IL, USA) and differences were considered significant at a p-value <0.05.
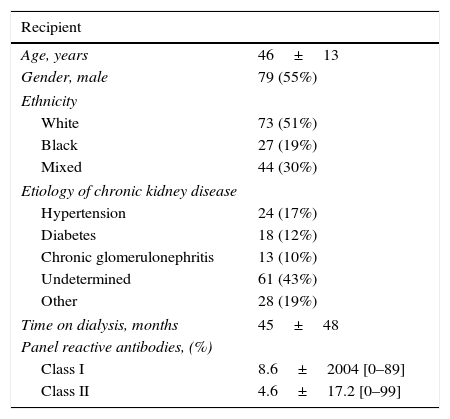
ResultsOf 155 screened patients, 144 patients were enrolled between March 2013 and August 2013. Eleven patients were excluded from this analysis, four due to missing donor CMV serologic information, six received alternative immunosuppressive regimens, and one was lost to follow up. Demographic characteristics of the study population (n=144) are shown in Table 1. The majority of the patients tested positive for CMV before transplantation (97%). Among high risk groups 3% were D+/R− and 39% received induction therapy.
Demographic characteristics of the study population (n=144).
| Recipient | |
|---|---|
| Age, years | 46±13 |
| Gender, male | 79 (55%) |
| Ethnicity | |
| White | 73 (51%) |
| Black | 27 (19%) |
| Mixed | 44 (30%) |
| Etiology of chronic kidney disease | |
| Hypertension | 24 (17%) |
| Diabetes | 18 (12%) |
| Chronic glomerulonephritis | 13 (10%) |
| Undetermined | 61 (43%) |
| Other | 28 (19%) |
| Time on dialysis, months | 45±48 |
| Panel reactive antibodies, (%) | |
| Class I | 8.6±2004 [0–89] |
| Class II | 4.6±17.2 [0–99] |
| Donor | |
|---|---|
| Age, years | 47±13 |
| Gender, male | 77 (53%) |
| Ethnicity | |
| White | 84 (59%) |
| Black | 15 (10%) |
| Mixed | 45 (31%) |
| Type | |
| Living | 32 (22%) |
| Decease | 112 (78%) |
| Standard | 65 (56%) |
| Expanded criteria donor | 47 (44%) |
| Cold ischemia time, hours | 20±6 (n=112) |
| Transplant | |
|---|---|
| CMV (IgG) | |
| D+/R− | 4 (3%) |
| D+/R+ | 132 (92%) |
| D-/R+ | 8 (5%) |
| HLA mismatches | 2.4±1.2 |
| Induction therapy | 56 (39%) |
| Immunosuppressive adjuvant drug | |
| Mycophenolate | 67 (46%) |
| Azathioprine | 77 (54%) |
CMV, cytomegalovirus; HLA, human leukocyte antigen; D, donor; R, recipient.
mean±standard deviation; range in brackets.
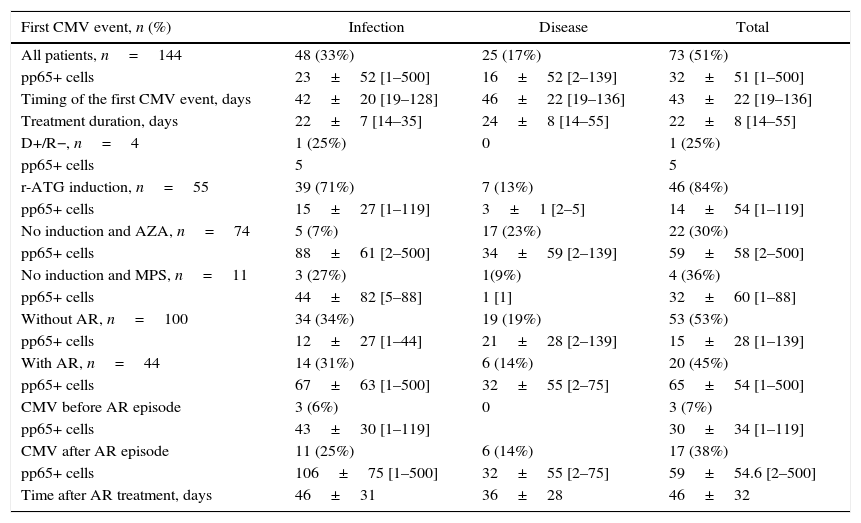
The overall incidence of first CMV infection/disease was 51% (73/144), 65% infection and 35% disease, with a global recurrence incidence of 23% (17/73), 71% (12/17) being recurrence infection and 29% (5/17) recurrence of disease. None of the patients developed tissue invasive disease. Mean time to first CMV event and the duration of treatment were similar among patients with infection or disease (Table 2).
Cytomegalovirus infection/disease.
| First CMV event, n (%) | Infection | Disease | Total |
|---|---|---|---|
| All patients, n=144 | 48 (33%) | 25 (17%) | 73 (51%) |
| pp65+ cells | 23±52 [1–500] | 16±52 [2–139] | 32±51 [1–500] |
| Timing of the first CMV event, days | 42±20 [19–128] | 46±22 [19–136] | 43±22 [19–136] |
| Treatment duration, days | 22±7 [14–35] | 24±8 [14–55] | 22±8 [14–55] |
| D+/R−, n=4 | 1 (25%) | 0 | 1 (25%) |
| pp65+ cells | 5 | 5 | |
| r-ATG induction, n=55 | 39 (71%) | 7 (13%) | 46 (84%) |
| pp65+ cells | 15±27 [1–119] | 3±1 [2–5] | 14±54 [1–119] |
| No induction and AZA, n=74 | 5 (7%) | 17 (23%) | 22 (30%) |
| pp65+ cells | 88±61 [2–500] | 34±59 [2–139] | 59±58 [2–500] |
| No induction and MPS, n=11 | 3 (27%) | 1(9%) | 4 (36%) |
| pp65+ cells | 44±82 [5–88] | 1 [1] | 32±60 [1–88] |
| Without AR, n=100 | 34 (34%) | 19 (19%) | 53 (53%) |
| pp65+ cells | 12±27 [1–44] | 21±28 [2–139] | 15±28 [1–139] |
| With AR, n=44 | 14 (31%) | 6 (14%) | 20 (45%) |
| pp65+ cells | 67±63 [1–500] | 32±55 [2–75] | 65±54 [1–500] |
| CMV before AR episode | 3 (6%) | 0 | 3 (7%) |
| pp65+ cells | 43±30 [1–119] | 30±34 [1–119] | |
| CMV after AR episode | 11 (25%) | 6 (14%) | 17 (38%) |
| pp65+ cells | 106±75 [1–500] | 32±55 [2–75] | 59±54.6 [2–500] |
| Time after AR treatment, days | 46±31 | 36±28 | 46±32 |
| Recurrent CMV events | Infection | Disease | Total |
|---|---|---|---|
| All patients, n=144 | 12 (8%) | 5 (3%) | 17 (11%) |
| pp65+ cells | 24±26 [2–119] | 18±15 [2–68] | 22±30 [2–119] |
| Timing after the first CMV event, days | 57±21 | 84±22 | 60±19 |
| Treatment duration, days | 25±9 | 24±2 | 23±9 |
Data are presented as mean±standard deviation and range in brackets.
pp65+ cells, number of positive cells/200.000 leucocytes.
CMV, cytomegalovirus; AR, acute rejection; r-ATG, rabbit anti-thymocyte globulin; AZA, azathioprine; MPS, mycophenolate sodium.
Of the 73 first episodes of CMV infection/disease, 56 (77%) occurred in high risk patients during preemptive therapy (1 D+/R−, 38 who received induction therapy, and 17 after treatment of acute rejection) and 17 (23%) in low risk patients (no induction, no D+/R−, no acute rejection) who had not received preemptive therapy.
Of the four patients in the high-risk D+/R− group one patient (25%) who also received induction therapy developed CMV infection. Of the 55 R+ patients who received induction therapy 38 (69%) developed CMV infection/disease before any treatment for acute rejection. Out of 37 patients treated for acute rejection (11 received induction therapy) 17 (46%) developed CMV infection/disease, seven after treatment with methylprednisolone and 10 after treatment with methylprednisolone and r-ATG. The incidence of CMV infection/disease after treatment for acute rejection was higher among patients who received induction therapy (8/11, 71%) compared to those without induction therapy (9/26, 35%). In low risk patients (R+, no induction, and no rejection), the incidence of CMV infection/disease was 28% (17/61).
There were 18 recurrent CMV infection/disease episodes, with 16 patients presenting one and one patient presenting two episodes. Recurrent episodes occurred in 14 patients who received induction therapy (five were treated for acute rejection before the first CMV episode) and in three patients that did not receive induction therapy but were treated for acute rejection before the first CMV episode. The number of pp65 positive cells tended to be higher in patients with infection versus disease, in patients receiving no induction, and in patients treated for acute rejection (Table 2).
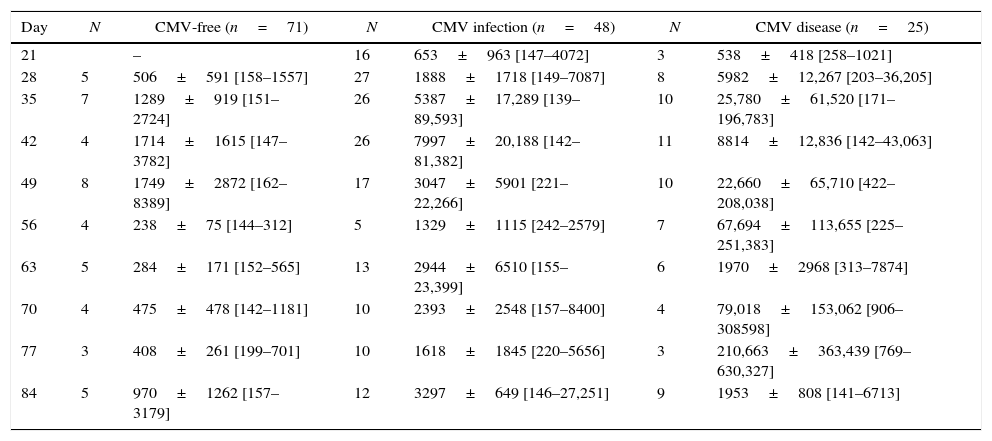
CMV viral replication by QNATData derived from blinded monitoring of CMV viral load by nucleic acid amplification test is shown in Table 3. The correlation between the number of pp65 positive cells and CMV copies/mL is expressed by an r2=0.633 (p<0.001). CMV viral replication was initially detected by week 3 after transplantation. High interindividual variability in CMV DNAemia was observed in patients developing CMV infection or disease. DNAemia tended to be higher among patients with CMV disease compared to infection.
CMV viral replication by quantitative nucleic acid amplification test (UI/mL).
| Day | N | CMV-free (n=71) | N | CMV infection (n=48) | N | CMV disease (n=25) |
|---|---|---|---|---|---|---|
| 21 | – | 16 | 653±963 [147–4072] | 3 | 538±418 [258–1021] | |
| 28 | 5 | 506±591 [158–1557] | 27 | 1888±1718 [149–7087] | 8 | 5982±12,267 [203–36,205] |
| 35 | 7 | 1289±919 [151–2724] | 26 | 5387±17,289 [139–89,593] | 10 | 25,780±61,520 [171–196,783] |
| 42 | 4 | 1714±1615 [147–3782] | 26 | 7997±20,188 [142–81,382] | 11 | 8814±12,836 [142–43,063] |
| 49 | 8 | 1749±2872 [162–8389] | 17 | 3047±5901 [221–22,266] | 10 | 22,660±65,710 [422–208,038] |
| 56 | 4 | 238±75 [144–312] | 5 | 1329±1115 [242–2579] | 7 | 67,694±113,655 [225–251,383] |
| 63 | 5 | 284±171 [152–565] | 13 | 2944±6510 [155–23,399] | 6 | 1970±2968 [313–7874] |
| 70 | 4 | 475±478 [142–1181] | 10 | 2393±2548 [157–8400] | 4 | 79,018±153,062 [906–308598] |
| 77 | 3 | 408±261 [199–701] | 10 | 1618±1845 [220–5656] | 3 | 210,663±363,439 [769–630,327] |
| 84 | 5 | 970±1262 [157–3179] | 12 | 3297±649 [146–27,251] | 9 | 1953±808 [141–6713] |
Mean±standard deviation (only values above 136UI/mL); range in brackets.
Among 71 patients who were not diagnosed with CMV infection/disease based on pp65 antigenemia test, 50 did not show any DNAemia and 21 (30%), 3 D+/R−, 8 who received induction therapy, 3 MPS and 10 AZA, presented asymptomatic transient DNAemia with up to 9000copies/mL during the first three months after transplantation (Table 3).
The receiver operating characteristic analysis (AUC=0.762±0.037, p<0.001) showed that CMV DNAemia ≥722UI/mL was associated with a sensitivity of 67% and 80% specificity to detect CMV infection diagnosed by pp65 antigenemia test. Likewise, the receiver operating characteristic analysis (AUC=0.849±0.042, p<0.001) showed that CMV DNAemia ≥2.169UI/mL was associated with a sensitivity of 61% and 85% specificity to detect CMV disease diagnosed by pp65 antigenemia test.
Efficacy and safetyThe incidence of first treated biopsy confirmed acute rejection was 26% and was higher among patients receiving AZA compared to MPS. Overall there were 48 treated acute rejection episodes, 3 graft losses, and 4 deaths during the first year after transplantation. Mean GFR at one month was 37.18±19.73ml/min with no difference comparing patients with or without CMV infection/disease (39.19±20.40 vs. 31.85±17.17ml/min, p=0.158), respectively. At 12 months mean GFR was lower in patients with CMV infection and/or acute rejection compared to patients without these events (Table 4).
Efficacy parameters during the first year after transplantation.
| Parameter | Total (n=144) |
|---|---|
| First treated BCAR | 37 (26%) |
| Incidence in patients receiving MPS, n=67 | 12 (18%) |
| Incidence in patients receiving AZA, n=77 | 25 (32%) |
| Time after transplant, days | 70±91 |
| Severity of the first BCAR* | |
| IA | 18 (48%) |
| IB | 18 (48%) |
| IIB | 1 (4%) |
| Treatment of 1st BCAR | |
| Methylprednisolone | 27 (73%) |
| Methylprednisolone+anti-thymocyte globulin | 10 (27%) |
| Patients with 2 treated acute rejection episodes | 4 |
| All treated acute rejection episodes | 48 |
| Severity of all treated acute rejection episodes* | |
| Tubulointerstitial nephritis | 2 (4%) |
| Borderline changes | 9 (19%) |
| IA | 18 (37.5%) |
| IB | 18 (37.5%) |
| IIB | 1 (2%) |
| Treatment of all acute rejection episodes | |
| Methylprednisolone | 38 (79%) |
| Methylprednisolone +anti-thymocyte globulin | 10 (21%) |
| Graft loss | 3 (2%) |
| Death | 4 (3%) |
| GFR at 12 months (ml/min) | |
| All patients (n=137) | 52.87±20.98 |
| With CMV infection/disease (n=68) | 45.92±17.87a |
| Without CMV infection/disease (n=69) | 58.46±21.02 |
| With acute rejection (n=36) | 47.09±18.28b |
| Without acute rejection (n=101) | 54.92±20.98 |
| With CMV infection/disease and with acute rejection (n=20) | 45.89±16.27 |
| With CMV infection/disease and without acute rejection (n=43) | 45.98±17.45 |
| Without CMV infection/disease and with acute rejection (n=20) | 46.72±18.09 |
| Without CMV infection/disease and without acute rejection (n=54) | 62.76±22.38c |
BCAR, biopsy confirmed acute rejection; MPS, mycophenolate sodium; AZA, azathioprine; GFR, glomerular filtration rate (MDRD formula); *Banff (2007).
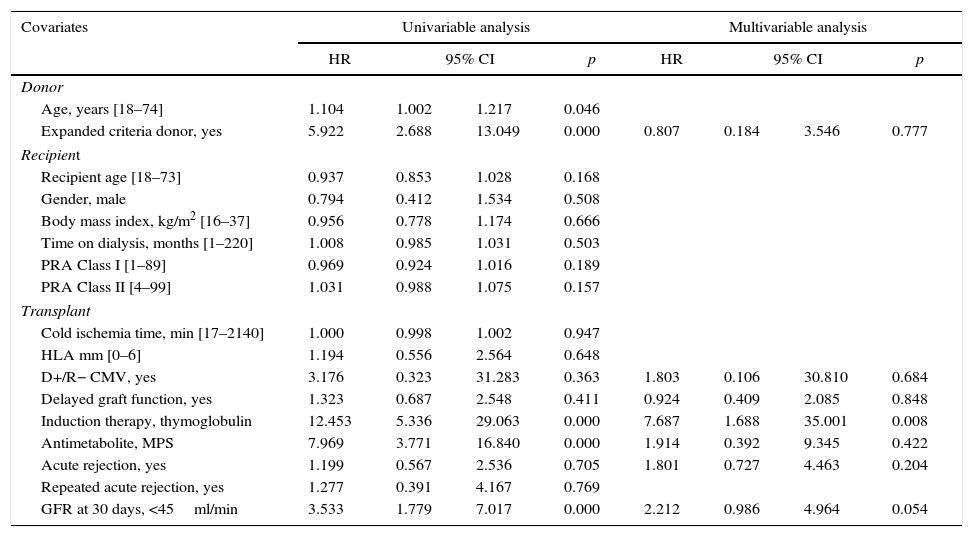
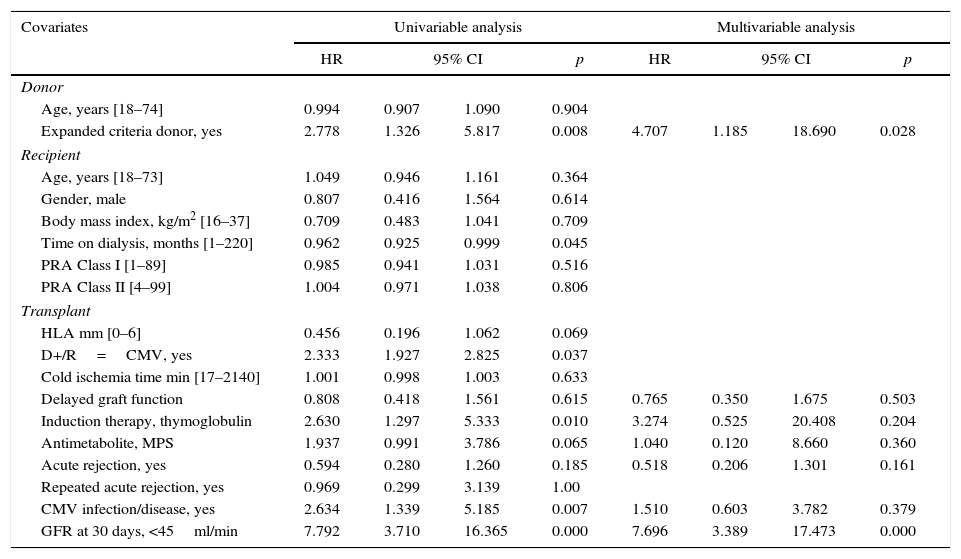
Donor age, expanded criteria donor, use of induction therapy, use of MPS, and renal function 30 days after transplantation were risk factors associated with CMV infection/disease. After multivariate analysis, only induction therapy remained independently associated with CMV infection/disease; renal function at 30 days reached marginal significance (p=0.054) (Table 5). Conversely, expanded criteria donor, time on dialysis, D+/R−, use of induction therapy, use of MPS, CMV infection/disease, and renal function 30 days after transplantation were risk factors associated with renal function 12 months after transplantation. After multivariate analysis, only expanded donor criteria and renal function 30 days after transplantation were independently associated with renal function 12 months after transplantation (Table 6).
Risk factors associated to CMV infection/disease during the first year after kidney transplantation.
| Covariates | Univariable analysis | Multivariable analysis | ||||||
|---|---|---|---|---|---|---|---|---|
| HR | 95% CI | p | HR | 95% CI | p | |||
| Donor | ||||||||
| Age, years [18–74] | 1.104 | 1.002 | 1.217 | 0.046 | ||||
| Expanded criteria donor, yes | 5.922 | 2.688 | 13.049 | 0.000 | 0.807 | 0.184 | 3.546 | 0.777 |
| Recipient | ||||||||
| Recipient age [18–73] | 0.937 | 0.853 | 1.028 | 0.168 | ||||
| Gender, male | 0.794 | 0.412 | 1.534 | 0.508 | ||||
| Body mass index, kg/m2 [16–37] | 0.956 | 0.778 | 1.174 | 0.666 | ||||
| Time on dialysis, months [1–220] | 1.008 | 0.985 | 1.031 | 0.503 | ||||
| PRA Class I [1–89] | 0.969 | 0.924 | 1.016 | 0.189 | ||||
| PRA Class II [4–99] | 1.031 | 0.988 | 1.075 | 0.157 | ||||
| Transplant | ||||||||
| Cold ischemia time, min [17–2140] | 1.000 | 0.998 | 1.002 | 0.947 | ||||
| HLA mm [0–6] | 1.194 | 0.556 | 2.564 | 0.648 | ||||
| D+/R− CMV, yes | 3.176 | 0.323 | 31.283 | 0.363 | 1.803 | 0.106 | 30.810 | 0.684 |
| Delayed graft function, yes | 1.323 | 0.687 | 2.548 | 0.411 | 0.924 | 0.409 | 2.085 | 0.848 |
| Induction therapy, thymoglobulin | 12.453 | 5.336 | 29.063 | 0.000 | 7.687 | 1.688 | 35.001 | 0.008 |
| Antimetabolite, MPS | 7.969 | 3.771 | 16.840 | 0.000 | 1.914 | 0.392 | 9.345 | 0.422 |
| Acute rejection, yes | 1.199 | 0.567 | 2.536 | 0.705 | 1.801 | 0.727 | 4.463 | 0.204 |
| Repeated acute rejection, yes | 1.277 | 0.391 | 4.167 | 0.769 | ||||
| GFR at 30 days, <45ml/min | 3.533 | 1.779 | 7.017 | 0.000 | 2.212 | 0.986 | 4.964 | 0.054 |
CMV, cytomegalovirus; HR, hazard ratio; CI, confidence intervals; PRA, panel reactive antibodies; HLA mm, histocompatibility leukocyte antigen mismatch; D, donor; R, recipient; MPS, mycophenolate sodium; GFR, glomerular filtration rate.
Risk factors associated with inferior renal function (GFR< 51.7ml/min) one year after kidney transplantation.
| Covariates | Univariable analysis | Multivariable analysis | ||||||
|---|---|---|---|---|---|---|---|---|
| HR | 95% CI | p | HR | 95% CI | p | |||
| Donor | ||||||||
| Age, years [18–74] | 0.994 | 0.907 | 1.090 | 0.904 | ||||
| Expanded criteria donor, yes | 2.778 | 1.326 | 5.817 | 0.008 | 4.707 | 1.185 | 18.690 | 0.028 |
| Recipient | ||||||||
| Age, years [18–73] | 1.049 | 0.946 | 1.161 | 0.364 | ||||
| Gender, male | 0.807 | 0.416 | 1.564 | 0.614 | ||||
| Body mass index, kg/m2 [16–37] | 0.709 | 0.483 | 1.041 | 0.709 | ||||
| Time on dialysis, months [1–220] | 0.962 | 0.925 | 0.999 | 0.045 | ||||
| PRA Class I [1–89] | 0.985 | 0.941 | 1.031 | 0.516 | ||||
| PRA Class II [4–99] | 1.004 | 0.971 | 1.038 | 0.806 | ||||
| Transplant | ||||||||
| HLA mm [0–6] | 0.456 | 0.196 | 1.062 | 0.069 | ||||
| D+/R=CMV, yes | 2.333 | 1.927 | 2.825 | 0.037 | ||||
| Cold ischemia time min [17–2140] | 1.001 | 0.998 | 1.003 | 0.633 | ||||
| Delayed graft function | 0.808 | 0.418 | 1.561 | 0.615 | 0.765 | 0.350 | 1.675 | 0.503 |
| Induction therapy, thymoglobulin | 2.630 | 1.297 | 5.333 | 0.010 | 3.274 | 0.525 | 20.408 | 0.204 |
| Antimetabolite, MPS | 1.937 | 0.991 | 3.786 | 0.065 | 1.040 | 0.120 | 8.660 | 0.360 |
| Acute rejection, yes | 0.594 | 0.280 | 1.260 | 0.185 | 0.518 | 0.206 | 1.301 | 0.161 |
| Repeated acute rejection, yes | 0.969 | 0.299 | 3.139 | 1.00 | ||||
| CMV infection/disease, yes | 2.634 | 1.339 | 5.185 | 0.007 | 1.510 | 0.603 | 3.782 | 0.379 |
| GFR at 30 days, <45ml/min | 7.792 | 3.710 | 16.365 | 0.000 | 7.696 | 3.389 | 17.473 | 0.000 |
CMV, cytomegalovirus; HR, hazard ratio; CI, confidence intervals; PRA, panel reactive antibodies; HLA mm, histocompatibility leukocyte antigen mismatch; D, donor; R, recipient; MPS, mycophenolate; GFR, glomerular filtration rate.
This study described the incidence and risk factors for CMV infection/disease in a cohort of kidney transplant recipients receiving no pharmacological prophylaxis. Furthermore, preemptive strategy was used only in patients deemed high risk for CMV infection/disease (D+/R−, use of r-ATG induction therapy, or after treatment of acute rejection). The overall incidence of CMV infection was 34% and CMV disease was 17% based on pp65 antigenemia. Importantly, of 73 patients in the first episode of CMV infection/disease, 17 (23%) presented at least one recurrent event, but no episodes of tissue invasive disease or late CMV infection (> 6 months) were observed. A recent study showed 33% recurrence rate and identified high viral load at initial diagnosis as independent risk factor.24 In our cohort of patients peak DNAemia ranged from 142 to 81.382UI/mL during CMV infection and from 142 to 630.327UI/mL during CMV disease.
The prevalence of the high risk D+/R− group (2.8%) was lower compared to that observed in Europe or North America. The incidence of CMV infection/disease in this group was only 25%. Nevertheless, in one larger prospective trial including 288 patients with similar demographic characteristics and receiving only preemptive therapy, the prevalence of the high risk D+/R− group was also low (5.2%), but the incidence of CMV infection/disease was 63%.25
The incidence of CMV infection/disease was 84% in patients receiving r-ATG induction therapy, confirming the results of previous studies.26,27 A recent meta-analysis showed that the risk of developing CMV infection/disease is 60% higher in patients receiving r-ATG induction therapy.28 Other reviews suggested that r-ATG induction therapy was associated with increased incidence of CMV infection/disease if prophylaxis were not used29 or when compared with basiliximab in low immunological risk patients.30
The incidence of CMV infection/disease was 48% (37/77) after treatment for acute rejection, confirming the need to restart preemptive therapy recommended by recent guidelines.2 A recent study showed that acute rejection was an independent risk factor associated with a 2.8-fold increased risk for tissue-invasive CMV disease within six months of kidney transplantation.31
On the other hand, in low risk patients (R+, no induction therapy, and no acute rejection) the incidence of CMV infection/disease was 28%. Furthermore, 15% (21/144) of the patients presented transient DNAemia (peak DNAemia ranging from 162 to 8389UI/mL) during the first three months after transplantation. In one study where patients were treated only based on clinical suspicion, transient DNAemia was observed in 53% of the patients.19 DNAemia tended to be higher in patients with disease compared to infection, perhaps due to early treatment prompted by preemptive therapy. All patients in this study were successfully treated with ganciclovir and no ganciclovir resistant mutations were detected. Therefore, none of these patients were treated with foscarnet.1
Using receiver operating characteristic analysis the optimal cutoff concentration for CMV disease was 2.169UI/mL, which is comparable to that observed in a cohort of CMV seropositive solid organ transplant recipients (3983UI/mL),32 in 97 liver transplant recipients (2000–5000copies/mL),33 and in 102 D+/R+ kidney transplant recipients receiving tacrolimus, mycophenolate and prednisone (1920IU/mL).19 On the other hand, higher cutoff values were proposed in other studies.34–36 This apparent discrepancy may be due to differences in ethnic background, donor/recipient pretransplant CMV serostatus, immunosuppressive regimens used, biological sample (plasma versus blood), and the QNAT used. This observation highlights the need to validate locally the strategy used to monitor CMV viral replication.
The recurrence rate of CMCV infection in our cohort of patients was 23% (17 of 73 patients with CMV infection/disease), which is lower than the 30% suggested in the last consensus (2). Of these 17 recurrent episodes, 12 (71%) were CMV infection and 5 (29%) CMV disease, suggesting that the preemptive strategy based on antigenemia test was adequate. The option to trigger treatment earlier based on the lower cutoff value of ≥722UI/mL for CMV infection is a matter of debate, considering the low proportion of patients with CMV recurrence who ultimately developed disease and the unnecessary treatment of those patients with low self-limited viral replication. Individual assessment, based on CMV viral replication kinetics and more frequent and prolonged monitoring is perhaps the ideal strategy, considering that recurrent episodes were diagnosed up to 106 days after completion of first treatment.
Among recognized risk factors associated with CMV infection/disease after transplantation, r-ATG induction was associated with an almost 8-fold increased risk compared to no induction. The representation of the high risk D+/R− group was too small to detect an association with CMV infection/disease. Interestingly, renal function at the end of the first month after transplant was associated with 2-times higher risk of development of CMV infection/disease. In a recent study, the prevalence of interstitial fibrosis and tubular atrophy at six weeks after transplantation was 3-fold higher in patients who experienced CMV infection later-on, compared to patients without CMV.37 In this study only a small difference in GFR measured at six weeks was observed comparing patient with or without CMV (57±23 vs. 61±23ml/min; p=0.083), similar to our observation.
Finally, it has been suggested that CMV infection/disease and acute rejection are associated with reduced renal function.1,38 In our cohort we were unable to find an independent association between CMV infection/disease or acute rejection and renal function at the end of the first year after transplantation. Instead, only expanded criteria donor and renal function at the end of first month were associated with inferior renal function at 12 months.
This study has some limitations, including the relative small sample size, low representation of the high risk D+/R− group, short follow up, unique demographic characteristics of the study population and immunosuppressive strategies used in a single center, and the findings may not be directly extrapolated to other kidney transplant recipients.
Strategies to manage CMV infection are evolving despite the growing use of universal prophylaxis. The high incidence of transient spontaneously resolved asymptomatic CMV viremia, the costs and adverse events associated with universal prophylaxis and late CMV infection after its discontinuation, and the uncertainties of the effect of CMV DNAemia on graft and patient outcomes are the driving reasons for the continued search for alternative strategies. Our data suggests that targeted preemptive therapy in patients with perceived higher risk for CMV infection/disease (D+/R−, use of r-ATG, treatment of acute rejection) is effective in preventing severe clinical presentation, including tissue invasive and late CMV infection. Furthermore, substitution of azathioprine and mycophenolate by mTOR inhibitors may further reduce the incidence and recurrence of CMV infection in patients receiving no pharmacological prophylaxis. Eventually, monitoring of CMV viral replication will be performed only in high risk patients and in those discontinuing the use of mTOR inhibitors.25 These effective strategies are associated with direct and indirect cost-savings and reduced use of human resources.
Conflicts of interestThe authors declare no conflicts of interest. This work was partially founded by Novartis.