Visceral Leishmaniasis is the most severe form of disease caused by the Leishmania donovani complex, with significant morbidity and mortality in developing countries. Worse outcomes occur among HIV-positive individuals coinfected with Leishmania. It is unclear, however, if there are significant differences on presentation between Visceral Leishmaniasis patients with or without HIV coinfection.
MethodsWe reviewed medical records from adult patients with Visceral Leishmaniasis treated at a reference healthcare center in Fortaleza – Ceará, Brazil, from July 2010 to December 2013. Data from HIV-coinfected patients have been abstracted and compared to non-HIV controls diagnosed with Visceral Leishmaniasis in the same period.
ResultsEighty one HIV-infected patients and 365 controls were enrolled. The diagnosis in HIV patients took significantly longer, with higher recurrence and death rates. Kala-azar's classical triad (fever, constitutional symptoms and splenomegaly) was less frequently observed in Visceral Leishmaniasis-HIV patients, as well as jaundice and edema, while diarrhea was more frequent. Laboratory features included lower levels of hemoglobin, lymphocyte counts and liver enzymes, as well as higher counts of blood platelets and eosinophils. HIV-infected patients were diagnosed mainly through amastigote detection on bone marrow aspirates and treated more often with amphotericin B formulations, whereas in controls, rK39 was the main diagnostic tool and pentavalent antimony was primarily used for treatment.
ConclusionsClinical and laboratory presentation of Visceral Leishmaniasis in HIV-coinfected patients may differ from classic kala-azar, and these differences may be, in part, responsible for the delay in diagnosing and treating leishmaniasis, which might lead to worse outcomes.
Leishmaniasis is the second most prevalent cause of protozoal diseases in the world and visceral presentation (VL or kala-azar) is the most severe form. World Health Organization (WHO) estimates 300 million new cases each year worldwide, with over 20,000 deaths.1 In Brazil, between 2001 and 2013, 46,642 cases were reported, and seven states (Ceará, Minas Gerais, Maranhão, Bahia, Piauí, Tocantins, and Pará) contributed with nearly 75% of notifications. In the same period, at least 3058 deaths due to the disease occurred throughout the country. Among VL patients, the number of cases reported in HIV-infected individuals has increased substantially. Between 2007 and 2013, 1602 cases of leishmaniasis in HIV-infected individuals were reported in Brazil.2
Most cases of VL in HIV-coinfected patients are caused by reactivation of a latent infection which becomes clinically apparent as the immunosuppression progresses and the parasite overcomes the contingency capacity of the host's immune system.3,4 Classic kala-azar, characterized by prolonged fever, constitutional symptoms, hepatosplenomegaly, cytopenias and hypergammaglobulinemia is not always manifested in immunocompromised individuals. In general, the clinical presentation is similar to the non-coinfected, although there is a tendency to involve organs and systems not usually parasitized, like lungs, skin, and gastrointestinal (GI) tract.5–7 It is unknown if potential differences in clinical and laboratory presentations might hinder the inclusion of VL in the differential diagnosis, potentially leading to delayed diagnosis and therapy, which might lead to more severe course of the disease and poorer outcomes.
In Brazil, Ceará is the state with more cases of kala-azar reported in the period of 2007–2013, with or without HIV-coinfection.2 Local data describing clinical features peculiar to coinfected individuals are scarce. Our goal was to identify such features in relation to monoinfected patients with VL, in order to alert clinicians about differences and similarities in clinical presentation that might expedite diagnosis and treatment initiation.
Material and methodsStudy designThis was a cross-sectional study in adult patients with VL and HIV-coinfection, aiming to describe their epidemiological, clinical and laboratory presentations at diagnosis, as well as the drug prescribed for therapy. All data gathered from coinfected subjects were compared to monoinfected VL controls.
Population and study placeMedical records of all patients with confirmed VL diagnosis treated at São José Infectious Diseases Hospital (Hospital São José de Doenças Infecciosas), in the period of July 2010 to December 2013 were reviewed. São José Hospital is the reference healthcare center for treatment of infectious diseases in the state of Ceará. Individuals under 18 years, patients not tested for HIV, and those who had prior diagnosis of leishmaniasis or had been previously treated with amphotericin B for any disease were excluded.
Data collectionEpidemiological data (age, gender, education level, and origin), clinical information (comorbidities, duration of symptoms, clinical symptoms at diagnosis), laboratory tests (complete blood count, biochemical blood tests, and diagnostic tests) and treatment of HIV-coinfected patients (VL-HIV group) were compared with HIV-negative controls (VL group) diagnosed at the center during the same period.
The symptoms were considered present if reported by the patient at diagnosis or verified on physical examination by the doctor who assisted the patient. In addition, signs or symptoms within the first 24h of hospitalization were considered, unless observed after the first dose of therapy. Regarding laboratory tests, only those collected immediately before the first dose of therapy were considered; for blood count, urea and creatinine (more sensitive to changes in medical interventions non-directly related with the treatment, such as intravenous hydration), we opted for the corresponding examination on arrival at the hospital or immediately previous to that moment.
Patients were treated at the discretion of their assisting physicians and most patients received treatment according to national guidelines at the time.
Diagnostic criteriaDiagnosis of HIV infection was performed in accordance to the Brazilian Health Ministry guidelines, and included Enzyme-Linked Immunosorbet Assays (ELISA), plus a confirmatory method (usually Indirect Immunofluorescence or Immunoblot). Immunochromatographic rapid tests were only considered as diagnostic if confirmed by one of the confirmatory method previously cited.
Visceral Leishmaniasis confirmation criteria included: (1) visualization of amastigotes in bone marrow aspirate or other clinical materials (except when detected exclusively on skin or nasal/oral mucous membranes); (2) serology (detection of anti-rK39 antibodies by immunochromatography or indirect immunofluorescence reaction in a title equal or greater than 1:80), associated with clinical and laboratory findings compatible with the disease.
StatisticsData were compiled in Microsoft Excel version 2007 and the statistical analyses were performed using Stata SE, version 11.2. Numerical variables were described as median and range and were compared using Mann–Whitney test. Categorical variables were described as frequencies and analyzed by Pearson's chi-square test or 2-tailed Fisher exact test. For statistical significance, p-value of less than 0.05 was admitted.
EthicsThe research project was approved by the Research Ethics Committee of São José Hospital (CAAE 26007713.3.0000.5044) and followed the national and international standards in clinical research.
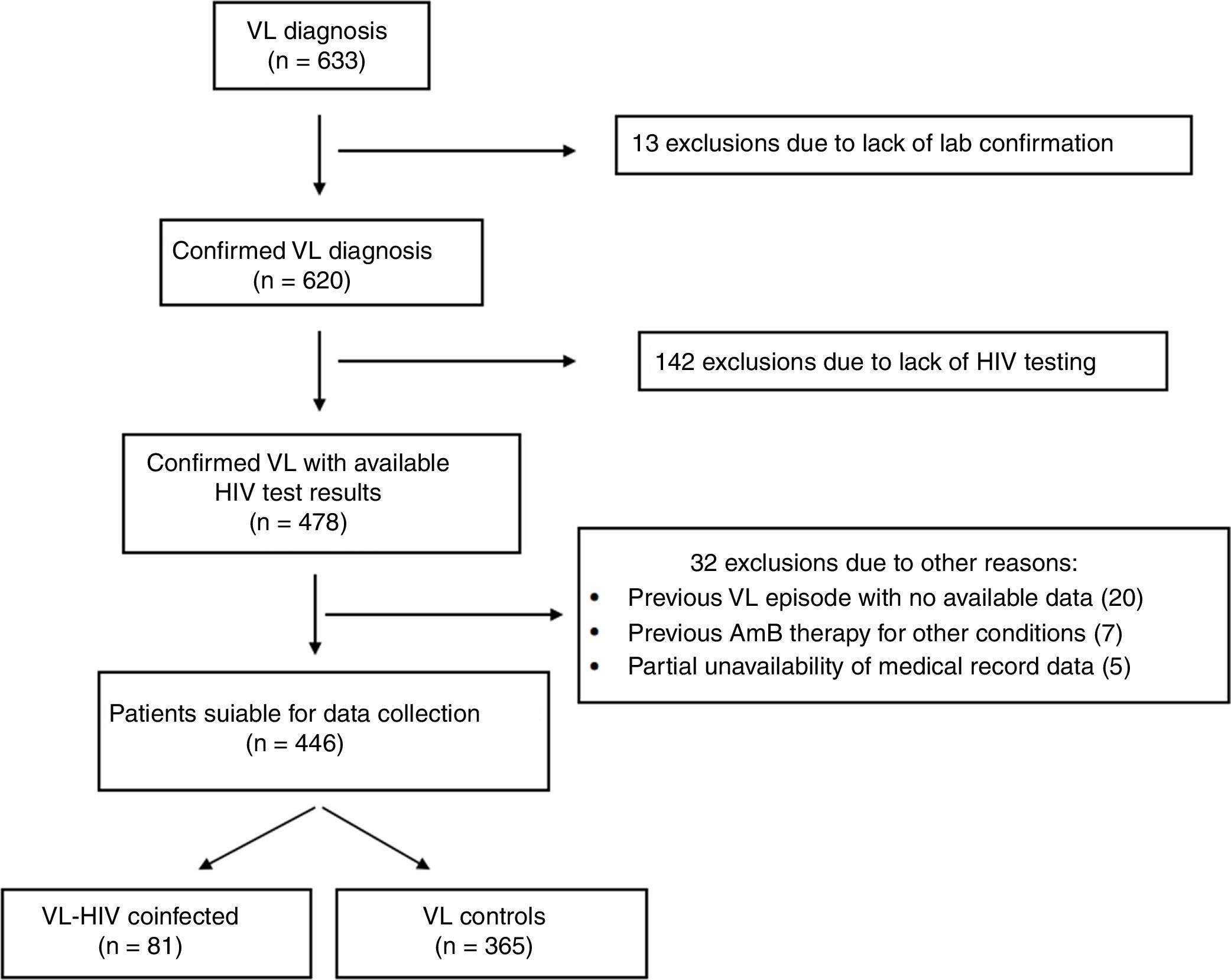
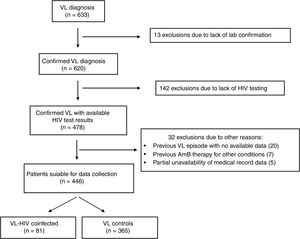
ResultsThe selection process is detailed in Fig. 1. Six hundred and thirty-three subjects were initially identified. A total of 187 subjects did not meet the eligibility criteria. Four hundred and forty-six subjects with VL – 81 coinfected with HIV and 365 non-coinfected – were included in the study.
Epidemiological dataThe median age of both groups was 38 years and the male gender was predominant (86.4% and 81.6% of the subjects on VL-HIV and VL groups, respectively). There were no differences concerning educational level, with 71.6% and 75.1% of the subjects having studied up to primary school. At the time of diagnosis, only two patients in each group lived outside Ceará. Approximately two thirds of individuals in each group lived in the capital city.
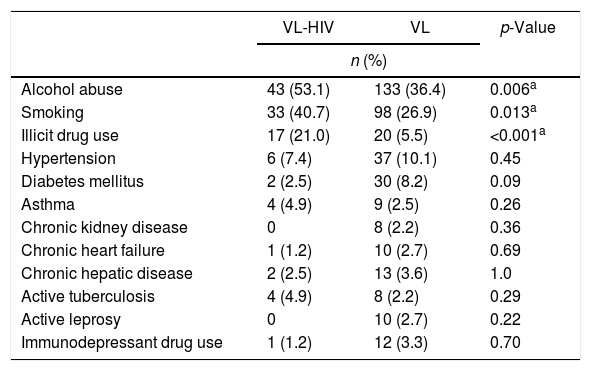
ComorbiditiesTable 1 shows habits and comorbidities at diagnosis. The most frequently used illicit drugs were cocaine, crack, and marijuana. Several patients reported combined use of these substances. Use of every substance was more prevalent in VL-HIV group (data not shown).
Comparison of habits and comorbidities of VL patients, according to HIV status.
| VL-HIV | VL | p-Value | |
|---|---|---|---|
| n (%) | |||
| Alcohol abuse | 43 (53.1) | 133 (36.4) | 0.006a |
| Smoking | 33 (40.7) | 98 (26.9) | 0.013a |
| Illicit drug use | 17 (21.0) | 20 (5.5) | <0.001a |
| Hypertension | 6 (7.4) | 37 (10.1) | 0.45 |
| Diabetes mellitus | 2 (2.5) | 30 (8.2) | 0.09 |
| Asthma | 4 (4.9) | 9 (2.5) | 0.26 |
| Chronic kidney disease | 0 | 8 (2.2) | 0.36 |
| Chronic heart failure | 1 (1.2) | 10 (2.7) | 0.69 |
| Chronic hepatic disease | 2 (2.5) | 13 (3.6) | 1.0 |
| Active tuberculosis | 4 (4.9) | 8 (2.2) | 0.29 |
| Active leprosy | 0 | 10 (2.7) | 0.22 |
| Immunodepressant drug use | 1 (1.2) | 12 (3.3) | 0.70 |
VL, Visceral Leishmaniasis.
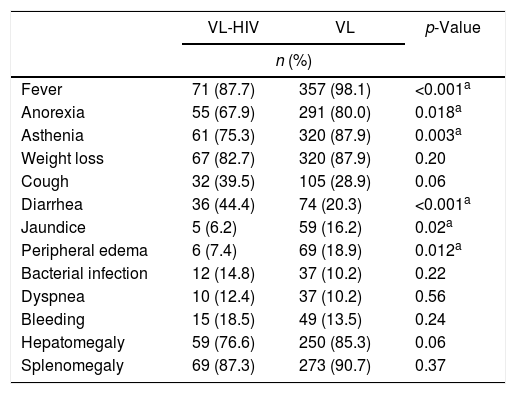
Table 2 depicts the clinical presentation of the patients at the time of diagnosis. Although there was no difference between groups in the frequency of bacterial superinfection, when each infection site was analyzed separately, there was significantly more skin and soft tissue infections in coinfected patients (p=0.04). The classic triad of VL (fever+constitutional symptoms+splenomegaly) was present more often in the VL group (86.7% vs. 75.9%, p=0.01). Median duration of symptoms attributable to the disease reported by patients also varied significantly between the groups. Patients in VL-HIV group had a median of three months (range, 0.4–24), versus two months in VL group (range, 0.2–12) (p=0.0001).
Comparison of clinical presentation of VL patients, according to HIV status.
| VL-HIV | VL | p-Value | |
|---|---|---|---|
| n (%) | |||
| Fever | 71 (87.7) | 357 (98.1) | <0.001a |
| Anorexia | 55 (67.9) | 291 (80.0) | 0.018a |
| Asthenia | 61 (75.3) | 320 (87.9) | 0.003a |
| Weight loss | 67 (82.7) | 320 (87.9) | 0.20 |
| Cough | 32 (39.5) | 105 (28.9) | 0.06 |
| Diarrhea | 36 (44.4) | 74 (20.3) | <0.001a |
| Jaundice | 5 (6.2) | 59 (16.2) | 0.02a |
| Peripheral edema | 6 (7.4) | 69 (18.9) | 0.012a |
| Bacterial infection | 12 (14.8) | 37 (10.2) | 0.22 |
| Dyspnea | 10 (12.4) | 37 (10.2) | 0.56 |
| Bleeding | 15 (18.5) | 49 (13.5) | 0.24 |
| Hepatomegaly | 59 (76.6) | 250 (85.3) | 0.06 |
| Splenomegaly | 69 (87.3) | 273 (90.7) | 0.37 |
VL, Visceral Leishmaniasis.
Note: One patient, who was disoriented and had no relatives with him on admission, had no clinical data. Not all patients had information about the presence or absence of visceromegaly at diagnosis.
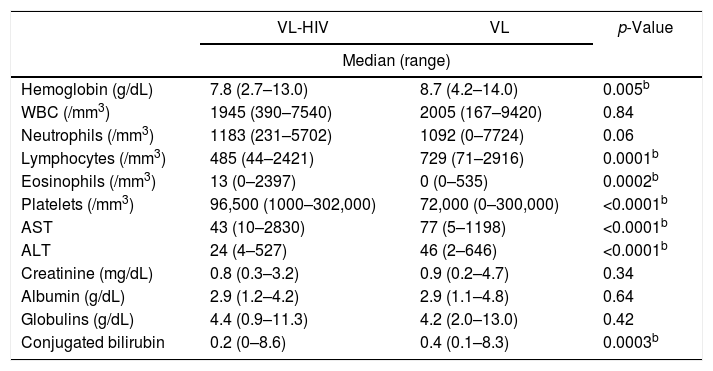
All results shown in Table 3 refer to time of diagnosis. Table 3 also shows the positivity of diagnostic tests. The positivity of bone marrow aspirate was better in the VL-HIV group, while the anti-rK39 proved superior in VL group (p<0.001 for both comparisons).
Comparison of laboratory and diagnostic tests of VL patients, according to HIV status.
| VL-HIV | VL | p-Value | |
|---|---|---|---|
| Median (range) | |||
| Hemoglobin (g/dL) | 7.8 (2.7–13.0) | 8.7 (4.2–14.0) | 0.005b |
| WBC (/mm3) | 1945 (390–7540) | 2005 (167–9420) | 0.84 |
| Neutrophils (/mm3) | 1183 (231–5702) | 1092 (0–7724) | 0.06 |
| Lymphocytes (/mm3) | 485 (44–2421) | 729 (71–2916) | 0.0001b |
| Eosinophils (/mm3) | 13 (0–2397) | 0 (0–535) | 0.0002b |
| Platelets (/mm3) | 96,500 (1000–302,000) | 72,000 (0–300,000) | <0.0001b |
| AST | 43 (10–2830) | 77 (5–1198) | <0.0001b |
| ALT | 24 (4–527) | 46 (2–646) | <0.0001b |
| Creatinine (mg/dL) | 0.8 (0.3–3.2) | 0.9 (0.2–4.7) | 0.34 |
| Albumin (g/dL) | 2.9 (1.2–4.2) | 2.9 (1.1–4.8) | 0.64 |
| Globulins (g/dL) | 4.4 (0.9–11.3) | 4.2 (2.0–13.0) | 0.42 |
| Conjugated bilirubin | 0.2 (0–8.6) | 0.4 (0.1–8.3) | 0.0003b |
| VL-HIV | VL | p-Value | |
|---|---|---|---|
| Positive/tested (%) | |||
| Amastigotes visualization in bone marrow | 56/67 (83.6) | 91/195 (46.7) | <0.001b |
| Anti-rK39 | 42/65 (64.6) | 333/343 (97.1) | <0.001b |
| Indirect immunofluorescence | 6/11 (54.5) | 5/6 (83.3) | 0.51 |
| Othera | 14 | 6 | – |
VL, Visceral Leishmaniasis; WBC, white blood cells; AST, alanine-aminotransferase; ALT, aspartate-aminotransferase.
When analyzing only patients who had been diagnosed with bone marrow examination, considering it as the gold standard, anti-rK39 sensitivity was significantly lower in HIV-infected individuals (48.7% versus 85.9% in the LV group, p<0.001).
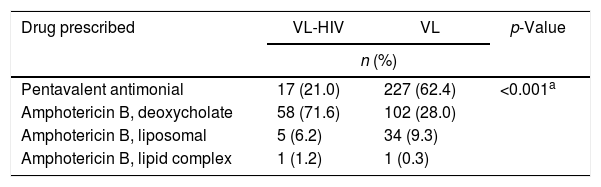
TreatmentData shown in Table 4 describe the initial drug prescribed. Treatment data refer only to the first drug initiated for treatment, not considering therapy switches.
Drugs used for treating VL patients, according to HIV status.
| Drug prescribed | VL-HIV | VL | p-Value |
|---|---|---|---|
| n (%) | |||
| Pentavalent antimonial | 17 (21.0) | 227 (62.4) | <0.001a |
| Amphotericin B, deoxycholate | 58 (71.6) | 102 (28.0) | |
| Amphotericin B, liposomal | 5 (6.2) | 34 (9.3) | |
| Amphotericin B, lipid complex | 1 (1.2) | 1 (0.3) | |
VL, Visceral Leishmaniasis.
Note: One patient on VL group died before starting treatment.
Despite having great epidemiological importance in the Brazilian context, the state of Ceará has few local published data about the features of VL in HIV-coinfected patients. Previous Brazilian studies have made significant contributions on this subject8–10; however, these studies have either enrolled a limited number of patients or used official database of mandatory disease notification. In this research, a relatively large number of patients were evaluated, both HIV-infected and -uninfected, and it seems clear that the presentation of VL in HIV-coinfected individuals may deviate from what is expected in a classical VL patient. This may lead to delayed diagnosis, since leishmaniasis may not be included in the differential diagnosis early on. This hypothesis may explain, at least in part, the longer duration of symptoms until the diagnosis VL was made in the HIV-infected group.
In our series, the classic symptoms were common in absolute terms in both groups, as described in other studies from Brazil, Ethiopia, India, and Spain.11–15 However, the classical clinical triad which has been used to define a suspected case of VL – fever, constitutional symptoms and splenomegaly – was found less frequently in the coinfected group, as well as jaundice and peripheral edema. These findings did not correlate with levels of albumin (similar in both groups) or with the magnitude of liver dysfunction. The less intense immune response against the parasite in HIV-infected individuals may be responsible for these differences. Dynamics of formation and maturation of hepatic granulomas in animals experimentally infected with viscerotropic species of Leishmania have been studied by some researchers.16,17 Cytokine profile generated by infected Kupffer cells and stimulated CD4 T lymphocytes seems to influence the quantity and quality of formed granulomas, which determines, to some extent, the level of inflammation generated. It is likely that, in individuals with impaired cellular immunity, such as in HIV-infected patients, less macrophage activation and lower levels of proinflammatory cytokines lead to formation of disorganized granulomas, causing poor control of parasite multiplication, along with less secondary injury of hepatocytes and intrahepatic bile canaliculi. This theory is supported by lower levels of aminotransferases and conjugated bilirubin found in our HIV coinfected patients. On systemic level, decreased production of certain macrophage activating cytokines in this population (e.g. IFN-y) may explain the lower frequency of fever and constitutional symptoms.18
On the other hand, diarrhea and cough, considered non-typical symptoms were more frequent in HIV-infected patients (although only diarrhea reached statistical significance). The common presence of these symptoms in the clinical picture of coinfected individuals can additionally mislead the diagnostic reasoning. They may represent Aids-related malabsorption syndrome, other opportunistic diseases, or greater parasitism in organs not regarded as important in classic kala-azar (especially digestive tract).13,19–22 We have no confirmation that there was greater parasite density in GI tract in our sample, but this finding is usually associated with a greater likelihood of relapse in HIV-infected individuals, as they are seen more often in association with lower T CD4+ cell counts.15,23
The longer duration of symptoms reported by our HIV-infected patients at the time of diagnosis may be due to less intense symptoms, not prompting these patients to seek medical care. On the other hand, health care teams may take longer to include kala-azar in the differential diagnosis, while investigating other opportunistic diseases that could have similar clinical presentation (e.g. disseminated histoplasmosis or mycobacteriosis). Whatever the reason, delay in diagnosing and appropriately treating these patients can lead to further spread of the parasite and, deepening of immunosuppression, may reduce the chance of long-term cure.
Hematologic results are clearly different in the three series. HIV-coinfected patients had significantly lower hemoglobin levels and lymphocyte counts at diagnosis. Most HIV patients diagnosed with VL had low T CD4+ counts at diagnosis and important hematopoietic disturbance due to HIV infection or other opportunistic diseases.8,15,24 Interestingly, unlike classical kala-azar, in which eosinophils are often absent from peripheral blood, in more than half of coinfected patients this cell was present in the initial hematologic evaluation. Platelet count, in turn, was higher in coinfected individuals, which could be related to different pathways concerning immune activation in a context of HIV infection.8,13,25
Unfortunately, molecular biology tests, which could increase the diagnostic accuracy, are unavailable in our center. Our service uses amastigotes visualization, primarily in bone marrow aspirate, and rapid anti-rK39 test as diagnostic tools. Because of its convenience, our internal protocol recommends performing the rapid test in patients who have suspected VL, and notifying and treating positive cases, if the patient presents clinical and laboratory findings compatible with the disease. This test is known as having good positive predictive value when clinical suspicion is high.26 However, the sensitivity of this test drops significantly in immunocompromised individuals, particularly in those infected with HIV.27,28 In this population, the performance of parasitological test was much better, perhaps because of the largest parasite density in the bone marrow, as previously suggested.29,30 Other factors, however, may have influenced this finding. In these patients, bone marrow aspiration is often used to investigate other diseases such as disseminated histoplasmosis (which is prevalent in our state and may have a very similar clinical and laboratory presentation as VL). Furthermore, it is possible that examiners spend more time analyzing smears of HIV-infected patients than of non-coinfected individuals. Some patients were diagnosed by finding the parasite incidentally in other tissues (e.g., buffy-coat when investigating disseminated histoplasmosis; lymph node, when investigating tuberculosis; biopsy samples of stomach and duodenum, after performing endoscopies when investigating persistent digestive symptoms). These findings have often surprised physicians in charge of patient care, because in most of these cases VL was not a strong diagnostic hypothesis.
As expected, clinicians prescribed preferentially amphotericin B rather than pentavalent antimonials when treating coinfected patients. Patients were treated according to guidelines of the Ministry of Health of Brazil, which considers antimony as the first-choice therapy for non-HIV-infected patients and recommends amphotericin B for those who are HIV-infected. Liposomal amphotericin B (AmBisome®, Gilead) had restricted indications and, because of its high cost, was prescribed only for patients who had failed treatment with deoxycholate amphotericin B or for those who developed renal failure.31 Preference for amphotericin B formulations for VL-HIV coinfection is based mostly on its superior in vitro potency against amastigotes, better outcomes in clinical trials when compared to antimonials, and potentially fatal toxicity reports (especially pancreatic) when using pentavalent antimonials on such patients.32–34 More recently, these recommendations have been modified and indications of liposomal amphotericin B were expanded, but most of the patients in our cohort were still treated according to the national recommendations prevailing at the time. Still, approximately one-fifth of coinfected individuals were inappropriately treated with antimonials.
Some limitations are noteworthy in this study. Epidemiological, clinical and laboratory data were collected from medical records. Although utmost care has been taken in the collecting process, some data (especially clinical) may not have been registered in the patient record. Other conditions (e.g. opportunistic infections) commonly found in advanced HIV disease were not analyzed and may have contributed with some of the differences described here. Regarding the study design, this was a descriptive study, and outcomes were not analyzed, as the focused on clinical and laboratory presentation. Finally, the center where the study was conducted is a reference center for infectious diseases, which may have increased the proportion of patients with severe disease, especially in the non-infected group. Nevertheless, the service still treats and monitors many patients with mild VL, and we believe that non-severe cases have indeed been well represented due to the relatively large sample size.
ConclusionOur results show significant differences in clinical and laboratory presentation of VL in HIV-coinfected patients when compared to the non-coinfected patients.
We consider that optimizing the recognition of the disease and starting specific therapy for leishmaniasis and HIV as early as possible could be relevant because they are modifiable factors. We propose that, in endemic areas for both diseases, clinicians assisting HIV-infected patients should increase their degree of clinical suspicion and include VL more often in the differential diagnosis of these patients, even if some of the classic clinical and laboratory features (like fever, splenomegaly or eosinopenia) are absent, and the immunoassay test is negative.
Conflicts of interestThe authors declare no conflicts of interest.
The authors are thankful to all healthcare workers from São José Hospital who collaborated with this study.