Successful treatment of HIV-positive children requires a high level of adherence (at least 95%) to highly active antiretroviral therapy. Adherence is influenced by factors related to the child and caregivers.
ObjectivesTo evaluate children and caregivers characteristics associated to children's adherence.
MethodsCross-sectional study, from September 2013 to June 2015, comprising a sample of caregivers of perinatally HIV-infected children, in the age group of 1–12 years, under antiretroviral therapy for at least 6 months and on follow-up in two AIDS reference centers in Salvador, Bahia. Caregiver self-reports were the sole source of 4 days adherence and sociodemographic information. Study participants who reported an intake >95% of prescribed medication were considered adherents. A variable, (“Composed Adherence”), was created to better evaluate adherence.
ResultsWe included 77 children and their caregivers. 88.3% of the caregivers were female, the median age was 38.0 years (IQR 33.5–47.5), 48.1% were white or mixed, 72.7% lived in Salvador and 53.2% had no fixed income. The 4 days child's adherence was associated only to caregivers that received less than a minimum salary (p<0.05), 70.1% of the caregivers had less than four years of formal education, 81.8% were children's relative and 53.2% of the caregivers were HIV positive. The caregiver's pharmacy refill, long-term adherence and 4 days adherence, were significantly associated with composed adherence (p<0.05). Child's long-term adherence was strongly associated to the 4 days child's adherence referred by caregiver (p<0.001).
ConclusionsOur results suggest the need of improvement in HIV-infected children adherence, through reinforcement of the caregivers own adherence.
The introduction of effective new antiretroviral therapies in the early 90s provided improved health and increased life expectancy for HIV-infected children.1,2 However, to achieve viral suppression, prevent disease progression and resistance to antiretroviral drugs, at least 95% adherence to therapy is required.3 Optimal adherence to therapy can be affected by several child-related factors, including biomedical, psychosocial, antiretroviral therapy and caregiver characteristics.4,5
Previous studies that assessed adherence to antiretroviral therapy in children showed that it varies from 57 to 81%, depending on the definition and the method by which adherence was verified. In two clinical protocols, which used the self-report method to evaluate adherence to therapy, full adherence was detected in 70–74% of the participants.6,7
Some caregiver's characteristics can affect children's adherence to treatment. In the case of mother-to-child transmission, death or severe disease of biological parents may unable them to take care of the child. This is especially true for mothers, who usually have to deal with the guilt associated with HIV transmission, and may have other comorbidities such as depression, which results in delayed child's care. In consequence, children care can be transferred to another family member, or even to institutions.8
The antiretroviral treatment is complex and requires constant changes in dosage according to the weight of the child, especially those who take syrups or powders that need previous reconstitution. Caregiver's low educational level may be related to non-adherence to treatment, due to the lack of prescription understanding. A Romanian study detected an association between poor adherence and low educational level of caregivers, which was also associated with treatment abandonment, in 40 cases of families with children on highly active antiretroviral therapy (HAART).9
It is remarkable that in resource-limited settings many HIV-infected children come from underprivileged families without financial conditions to provide proper food supply.10 This may be related with caregiver's decision to skip a dose of a specific ARV drug, when the child was not fed, because it was recommended to be administered with food, or after a meal.10 Lack of enough money to pay for transportation to the pharmacy for antiretroviral drugs withdrawal, can be also a cause of non-adherence.10–13
The main objective of this study was to evaluate the relationship between different caregivers characteristics (foster parents, biological parents, and relatives), and children's adherence by using two different approaches: child's “composed adherence” and the 4-day child's adherence, as reported by caregivers.
Material and methodsStudy settingThis study was conducted at CEDAP (Diagnostic Center, for assistance and research in HIV) and HUPES (Professor Edgard Santos University Hospital), two referral centers for care of HIV-infected children, in Salvador, Brazil. CEDAP is a public service, composed by a multidisciplinary team, which provides assistance to individuals with HIV/AIDS. The HUPES, is a large tertiary hospital that includes both, inpatient and outpatient AIDS clinics.
Study design and populationThis cross-sectional study was conducted from September 2013 to June 2015. The inclusion was restricted to HIV perinatally infected children, aged one to 12 years old, under antiretroviral therapy for at least six months and regular care at HUPES or CEDAP, and to their caregivers. All individuals in charge of routine administration of antiretroviral drugs to enrolled children were identified as caregivers.
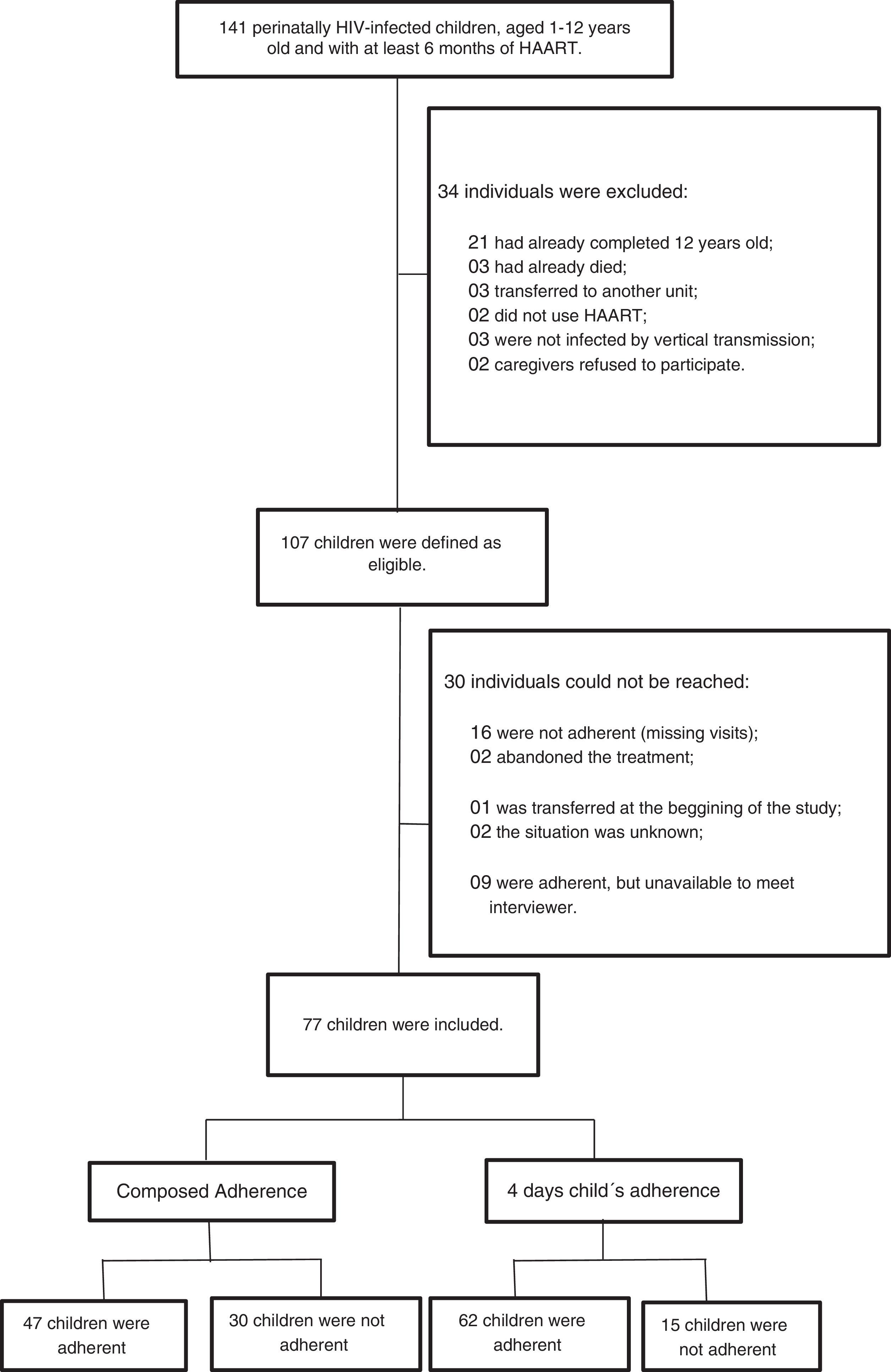
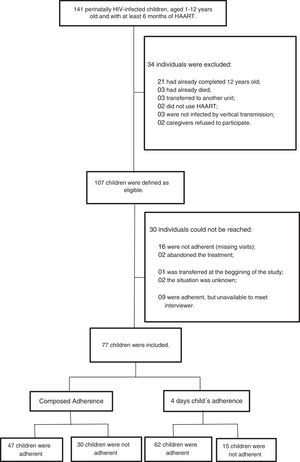
A total of 141 children were under care in the two-referral centers. Thirty-four of them were excluded: 21 (14.9%) were older than 12 years, three (2.1%) died, three (2.1%) were transferred to another reference center, two (1.4%) were not on HAART, three were not infected by vertical transmission, and two (1.41%) caregivers refused to participate.
From the 107 eligible children, 30 participants were excluded: 16 were not adherent to the treatment (irregular ART pharmacy refill and persistently high viral load), nine were not available to meet the investigators, two had abandoned treatment, one was transferred to another reference center at the beginning of the study, and the remaining two could not be contacted. Thus, 77 (72.0%) children were evaluated (Fig. 1).
Questionnaire design and adherence assessmentCaregiver demographic characteristicsCaregivers were asked to answer a structured questionnaire with sociodemographic information: relationship with the child (parent, relative, or adoptive caregiver), caregiver HIV status, age, gender, residence (state capital city or countryside), race/ethnicity (white/mixed or black), functional status (employed, self-employed, unemployed, housewife, retired, or pensioner), family's monthly income (no household income/less than one Minimum Salary or one to five minimum salaries), formal education (no school/primary school or secondary school/Higher education), use of alcohol or illicit drugs, and disclosure of HIV status to at least one member of the family.
Electronic medical chart reviewMedical charts were reviewed to obtain the most recent HIV viral load, CD4+ lymphocyte count, and ARV regimen. Pharmacy refill records were collected for all antiretroviral medications prescribed in the last six months.
Adherence to ARV medicationCaregiver's self-reports were the sole source of the 4-day adherence data. For the analysis of adherence, the “Adherence Follow-up Questionnaire”, previously developed by the Aids Clinical Trial Group (ACTG) and drawn up originally in English,14 was carefully translated and adapted to the local language, into two questionnaires, one for evaluation of children's adherence and the other for adherence of HIV infected caregivers.
In both adherence questionnaires (caregivers and children), caregivers were asked to describe the ART regimen, to report on the number of missed doses for each medication per day, during the past four days. In addition, caregivers were asked the last time the medications were missed (past week, past 1–4 weeks, past month, past 1–3 months, >3 months ago) as a measure of “long-term adherence”.
Adherence for last four days, was calculated as the number of doses taken in the last four days/total number of prescribed doses for those four days. One dose was defined as any administration of each drug, regardless of the amount of pills. If the individual takes three medications and each one of them is twice a day, it was considered six doses per day. Study participants who reported an intake of ≥95% of prescribed medication were considered adherent and those with a reported intake of <95% were classified as non-adherent. A question regarding compliance with specific instructions for a given drug and how closely these instructions were followed was also included in the survey.
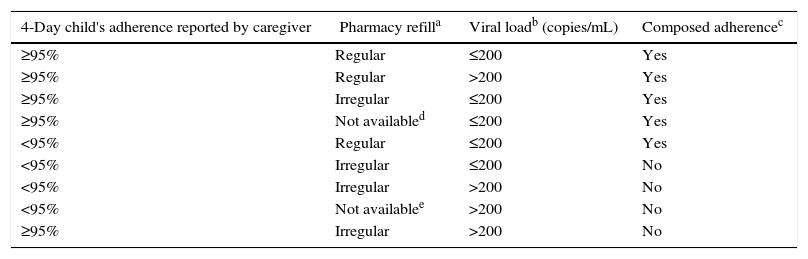
“Composed Adherence” was defined as the combination of three adherence measures: 4-day child's adherence reported by caregiver (≥95% or <95%), pharmacy refill in the last six months (regular if antiretrovirals were taken in the last 6 months without missing), and viral load (≤ 200 or >200copies/mL). Tables 1 and 2 summarizes these definitions.
Criteria to define children as “adherent” or “non-adherent” according to composed adherence method, in Salvador-Brazil, 2013–2015.
| 4-Day child's adherence reported by caregiver | Pharmacy refilla | Viral loadb (copies/mL) | Composed adherencec |
|---|---|---|---|
| ≥95% | Regular | ≤200 | Yes |
| ≥95% | Regular | >200 | Yes |
| ≥95% | Irregular | ≤200 | Yes |
| ≥95% | Not availabled | ≤200 | Yes |
| <95% | Regular | ≤200 | Yes |
| <95% | Irregular | ≤200 | No |
| <95% | Irregular | >200 | No |
| <95% | Not availablee | >200 | No |
| ≥95% | Irregular | >200 | No |
Pharmacy refill was regular when the caregiver took the ARVs for the child during the previous 6 months without missing and irregular. When the caregiver did not took the ARVs for the child during the previous 6 months at least 1 month.
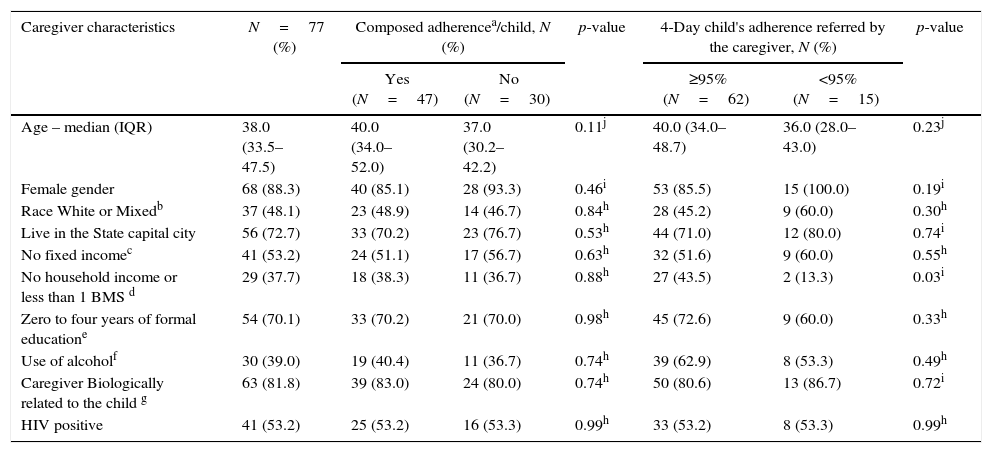
Sociodemographic characteristics of the caregiver, and the association with composed adherence and 4-day criteria, in Salvador-Brazil, 2013–2015.
| Caregiver characteristics | N=77 (%) | Composed adherencea/child, N (%) | p-value | 4-Day child's adherence referred by the caregiver, N (%) | p-value | ||
|---|---|---|---|---|---|---|---|
| Yes (N=47) | No (N=30) | ≥95% (N=62) | <95% (N=15) | ||||
| Age – median (IQR) | 38.0 (33.5–47.5) | 40.0 (34.0–52.0) | 37.0 (30.2–42.2) | 0.11j | 40.0 (34.0–48.7) | 36.0 (28.0–43.0) | 0.23j |
| Female gender | 68 (88.3) | 40 (85.1) | 28 (93.3) | 0.46i | 53 (85.5) | 15 (100.0) | 0.19i |
| Race White or Mixedb | 37 (48.1) | 23 (48.9) | 14 (46.7) | 0.84h | 28 (45.2) | 9 (60.0) | 0.30h |
| Live in the State capital city | 56 (72.7) | 33 (70.2) | 23 (76.7) | 0.53h | 44 (71.0) | 12 (80.0) | 0.74i |
| No fixed incomec | 41 (53.2) | 24 (51.1) | 17 (56.7) | 0.63h | 32 (51.6) | 9 (60.0) | 0.55h |
| No household income or less than 1 BMS d | 29 (37.7) | 18 (38.3) | 11 (36.7) | 0.88h | 27 (43.5) | 2 (13.3) | 0.03i |
| Zero to four years of formal educatione | 54 (70.1) | 33 (70.2) | 21 (70.0) | 0.98h | 45 (72.6) | 9 (60.0) | 0.33h |
| Use of alcoholf | 30 (39.0) | 19 (40.4) | 11 (36.7) | 0.74h | 39 (62.9) | 8 (53.3) | 0.49h |
| Caregiver Biologically related to the child g | 63 (81.8) | 39 (83.0) | 24 (80.0) | 0.74h | 50 (80.6) | 13 (86.7) | 0.72i |
| HIV positive | 41 (53.2) | 25 (53.2) | 16 (53.3) | 0.99h | 33 (53.2) | 8 (53.3) | 0.99h |
p<0.05 for all comparisons. PI, protease inhibitor; IQR, interquartile range.
The main outcome variable of this study was children's adherence. There were two definitions of adherence: composed adherence and the 4-day children's adherence reported by caregivers. The median values with interquartile ranges (IQR) were used as measures of central tendency because continuous variables (caregivers's age, children's age, treatment duration, and log10 HIV viral load) were not normally distributed. The comparison between these variables and study groups (composed adherence and 4-day child's adherence) was also performed, by using the Mann–Whitney U-test. The Fisher's or chi-square tests were used to determine if categorical variables were significantly associated with adherence. A p-value below 0.05 was considered statistically significant. Statistical analyses were performed by using SPSS 20.0 (IBM statistics).
Ethical considerationsThe present study was approved by the Health Research Ethics Committee of Climério de Oliveira Maternity, in August 2013 (approval decision number 376.534).
ResultsSociodemographic characteristics of caregivers and their association with the “composed adherence” and the 4-day children's adherence (reported by caregivers) are shown in Table 2. Among the 77 caregivers interviewed, 88.3% were female, the median age was 38.0 years (IQR 33.5–47.5), 48.1% declared themselves as white or racially mixed, 72.7% lived in Salvador and more than a half (53.2%) had no fixed income. Family income of less than a minimum wage (37.7%) was associated to 4-day children's adherence ≥95%, as reported by caregivers (p<0.05).
Regarding caregiver's characteristics, 70.1% of them had less than four years of formal education, 39.0% reported alcohol use, 81.8% were children's relatives (parents, grandparents, uncles, aunts) and 53.2% were HIV infected. Only one caregiver was drug user.
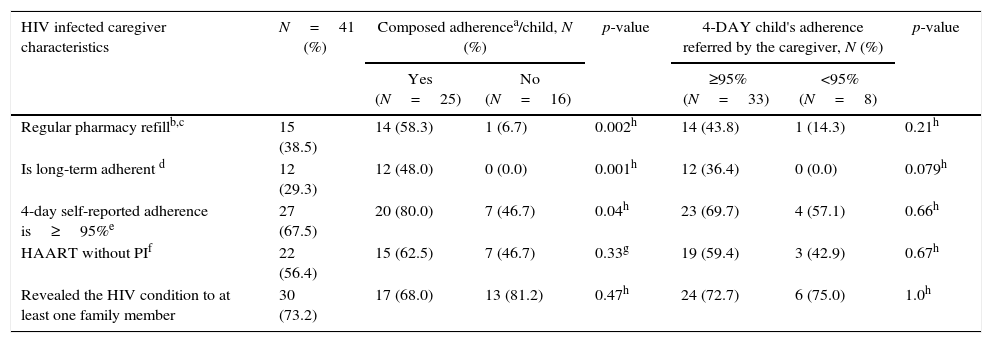
As expected, the adherence factors of the HIV-infected caregivers (caregiver's pharmacy refill, long term adherence and 4-day self-reported adherence) were associated (p<0.05) to child's “composed adherence”. Among the 41 HIV infected caregivers, 15 (38.5%) were regular on pharmacy refill. Of these, 14 (93.3%) had children who fulfilled composed adherence criteria. Furthermore, all children cared by caregivers with long-term adherence (12) were adherent to therapy, while among those cared by 27 caregivers that had 4-day self-reported adherence≥95%, only 20 (74.0%) were adherent, according to composed adherence criteria. Most (56.4%) of the HIV-infected caregivers were on non-nucleoside based HAART, and 73.2% of them had disclosed their HIV serostatus to at least one family member (Table 3).
HIV infected caregiver characteristics and the association with composed adherence and 4-day adherence criteria, in Salvador-Brazil, 2013–2015.
| HIV infected caregiver characteristics | N=41 (%) | Composed adherencea/child, N (%) | p-value | 4-DAY child's adherence referred by the caregiver, N (%) | p-value | ||
|---|---|---|---|---|---|---|---|
| Yes (N=25) | No (N=16) | ≥95% (N=33) | <95% (N=8) | ||||
| Regular pharmacy refillb,c | 15 (38.5) | 14 (58.3) | 1 (6.7) | 0.002h | 14 (43.8) | 1 (14.3) | 0.21h |
| Is long-term adherent d | 12 (29.3) | 12 (48.0) | 0 (0.0) | 0.001h | 12 (36.4) | 0 (0.0) | 0.079h |
| 4-day self-reported adherence is≥95%e | 27 (67.5) | 20 (80.0) | 7 (46.7) | 0.04h | 23 (69.7) | 4 (57.1) | 0.66h |
| HAART without PIf | 22 (56.4) | 15 (62.5) | 7 (46.7) | 0.33g | 19 (59.4) | 3 (42.9) | 0.67h |
| Revealed the HIV condition to at least one family member | 30 (73.2) | 17 (68.0) | 13 (81.2) | 0.47h | 24 (72.7) | 6 (75.0) | 1.0h |
p<0.05 for all comparisons. PI, protease inhibitor.
Table 4 shows sociodemographic and treatment characteristics of children that were associated to “Composed adherence”, and 4-day child's adherence reported by caregivers. Most of the children (54.5%) were male, median age was 94.2 months (IQR 58.7–118.5). Regarding children's antiretroviral regimens, 41.6% were using a combination of liquid drugs and pills, and most of children were adherent (44.7%), according to composed adherence. No association was found between these factors. Children's adherence, as reported by caregivers, was high, with 80.5% of children presenting adherence higher than 95%.
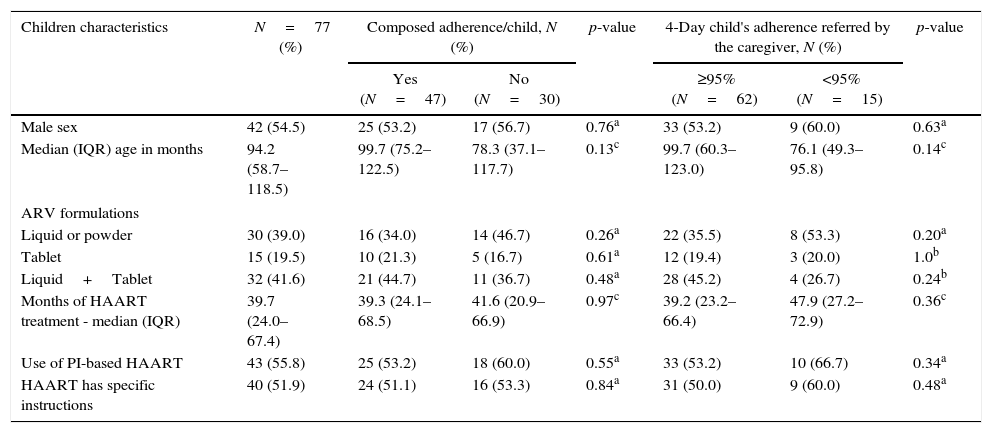
Children characteristics and the association to composed adherence and 4-day adherence criteria, in Salvador-Brazil, 2013–2015.
| Children characteristics | N=77 (%) | Composed adherence/child, N (%) | p-value | 4-Day child's adherence referred by the caregiver, N (%) | p-value | ||
|---|---|---|---|---|---|---|---|
| Yes (N=47) | No (N=30) | ≥95% (N=62) | <95% (N=15) | ||||
| Male sex | 42 (54.5) | 25 (53.2) | 17 (56.7) | 0.76a | 33 (53.2) | 9 (60.0) | 0.63a |
| Median (IQR) age in months | 94.2 (58.7–118.5) | 99.7 (75.2–122.5) | 78.3 (37.1–117.7) | 0.13c | 99.7 (60.3–123.0) | 76.1 (49.3–95.8) | 0.14c |
| ARV formulations | |||||||
| Liquid or powder | 30 (39.0) | 16 (34.0) | 14 (46.7) | 0.26a | 22 (35.5) | 8 (53.3) | 0.20a |
| Tablet | 15 (19.5) | 10 (21.3) | 5 (16.7) | 0.61a | 12 (19.4) | 3 (20.0) | 1.0b |
| Liquid+Tablet | 32 (41.6) | 21 (44.7) | 11 (36.7) | 0.48a | 28 (45.2) | 4 (26.7) | 0.24b |
| Months of HAART treatment - median (IQR) | 39.7 (24.0–67.4) | 39.3 (24.1–68.5) | 41.6 (20.9–66.9) | 0.97c | 39.2 (23.2–66.4) | 47.9 (27.2–72.9) | 0.36c |
| Use of PI-based HAART | 43 (55.8) | 25 (53.2) | 18 (60.0) | 0.55a | 33 (53.2) | 10 (66.7) | 0.34a |
| HAART has specific instructions | 40 (51.9) | 24 (51.1) | 16 (53.3) | 0.84a | 31 (50.0) | 9 (60.0) | 0.48a |
PI, protease inhibitor; IQR, interquartile range.
Among children included in the study, the median treatment time was 39.7 months (IQR 24.0–67.4), more than a half (55.8%) were using protease inhibitor-based HAART, and 51.9% of them reported to have received specific instructions from medical doctors, on how to use their ARV regimen. Median CD4+ cell count was 1064cell/mm3 (IQR 793.5–1450.5). Most children (93.5%) were followed up in CEDAP.
Table 5 shows the association between children's treatment and 4-day children's adherence reported by caregivers. Long-term adherence was strongly associated to 4-day children's adherence, as reported by caregivers (p<0.001).
Children characteristics and the association to composed adherence and 4-day adherence criteria, in Salvador-Brazil, 2013–2015.
| Children characteristics | N=77 (%) | 4-Day child's adherence referred by the caregiver, N (%) | p-value | |
|---|---|---|---|---|
| ≥95% (N=62) | <95% (N=15) | |||
| Median (IQR) of the Log10 viral loada | 0.0 (0.0–3.7) | 0.0 (0.0–3.2) | 3.3 (0.0–4.0) | 0.16e |
| The child is long-term adherentb | 40 (51.9) | 39 (62.9) | 1 (6.7) | 0.000d |
| Irregular pharmacy refillc | 44 (58.7) | 33 (54.1) | 11 (78.6) | 0.13d |
p<0.05 for all comparisons. IQR, interquartile range.
There are different methods to assess children's antiretroviral adherence. They include direct methods such as biologic markers and body-fluids assays, or indirect methods such as self-report, caregivers-reports, pill count, pharmacy records, and electronic drug monitoring (such as MEMS caps).15 An important feature of our study is that we combined one biologic marker (viral load), caregiver reports, and pharmacy refill records to produce one adherence composite, and compared this approach with a traditional method (caregiver reports).
In one open-label study of HAART conducted in the United States and Puerto Rico, combinations of at least two methods (self-report and clinic attendance) to produce an adherence evaluation tool were used.15 In developing countries caregiver reports are commonly employed to assess children's adherence, but there are no gold standard methods for measuring it.15–18 Results from seven studies that used caregivers reports, showed that 34–100% of caregivers reported 100% adherence and some of them showed that caregivers generally overestimate adherence in comparison with other methods.5,6,19–23
Other results on caregiver characteristics indicated that children are more adherent if they receive their medications from foster parents rather than the biological ones, or other relatives.7,24 We did not find associations between these variables (p=0.23). A Brazilian cross-sectional study also evaluated the impact of caregivers’ factors on children's adherence and concluded that children's non-adherence was significantly associated with caregivers low-educational level and to absence of biological parents.25
In this study, we detected an opposite association: the large majority of the caregivers had no income or received less than a minimum wage, but children under their care had a high rate of optimal adherence (≥95%), in the 4-day caregiver report. A reasonable explanation for this result relies on the fact that caregivers having a better monthly income, probably have a regular work, and would have less available time to care for the children under treatment. On the other hand, several studies indicate that financial constraint can be an obstacle to maintain child's adherence and collect medication on time. Some of the reasons are the distance barrier or lack of access to easy transportation to HIV clinic, forcing patients/caregivers to choose between paying for transportation or using the money for food.10,26,27 Another potential explanation for this discrepancy could be the low accuracy of such evaluations, for different populations.
An observational cross-sectional study conducted in Brazil evaluated 108 children and adolescents. Adherence was assessed using a questionnaire and pharmacy refill register. Patients and/or their caregivers were asked about the administration of prescribed medication in the last 24h or last seven days. The prevalence of non-adherence was 45.4% based on pharmacy refill and the authors concluded that this method has a high sensitivity to detect non-adherence.28
It is known that caregiver factors are important to pediatric adherence, which is directly or indirectly influenced by the resources and efficacy of care provided by their caregivers. A qualitative study conducted with 71 maternal caregivers of HIV infected children, administered an open-ended interview on questions concerning caregiving activities. Seventeen percent of the mothers with HIV, provided information about their own non-adherence. The low rate of mothers’ adherence in that study negatively influenced their children's adherence.29
In a multicenter, comparative trial, children older than 10 years were found to have better adherence than younger children, although the number of children in this group was small and most of them younger than 12 years.6 Our results did not detect any association between these two variables (composed adherence and 4-day child's adherence referred by the caregiver ≥95%).
Another challenging aspect for pediatric patients’ adherence is the administration of the correct dose of each ARV drug, due to limited availability of fixed-dose formulations, and differences in the way drugs are metabolized. Combining different drugs in appropriate formulations is particularly complex for infants and children, and often it results in under or overdosing. Moreover, dividing pills into halves or quarters does not ensure adequate dosing for children.30 As showed in this study, better adherence was observed in children taking pills only, compared to liquid formulations. Although this association did not reach statistical significance, it was probably due to the small sample size.
In a prospective cohort study, ART adherence levels and the ability of adherence measures to predict viral suppression among HIV infected children were evaluated. Regression modeling indicated that adherence rate was inversely, but not significantly, associated with log viral load at enrollment or 6-month visit, similarly to our findings.31 There was a significant association between long-term and 4-day children's adherence, as referred by caregiver. Long-term adherence in our study means that the child never missed a dose, according to caregiver's report, and could be defined as perfect adherence.
The limitations of our study include the cross-sectional design, which did not allow for the evaluation of adherence over time, the use of caregivers’ self-report adherence that can overestimate the true prevalence of adherence, and the small sample size, which limited our ability to investigate interactions among study variables by multivariate analysis.
However, a strong component of this study is the composed adherence, which showed that, the 4-day child's adherence referred only by the caregiver, could not be trusted as the only way to assess adherence, as used in other studies. This adherence evaluation tool demonstrated to be stronger than other methods used before. It is easy to apply and requires only a biological marker (plasma HIV-1 viral load), in association to pharmacy records and a self-reported questionnaire. Further studies are required to validate and confirm the usefulness of this tool.
Taken together, our results suggest the need of adherence improvement in HIV infected children, especially those who depend on caregivers. It is clear that some caregiver's characteristics can influence children's adherence. Actions focused on caregivers’ education and reinforcement of the need of correct use of antiretrovirals are urgently needed in order to improve outcomes of HIV infected children.
Conflicts of interestThe authors declare no conflicts of interest.