We sought to characterize the antibiotic susceptibility of strains of Stenotrophomonas maltophilia isolated from clinical samples, and the role of Stenotrophomonas maltophilia biofilm in antibiotic resistance.
MethodsFifty-one clinical Stenotrophomonas maltophilia isolates were obtained from patients with nosocomial infection in the surgical wards and ICUs of six general hospitals in Tianjin, China. In vitro models of Stenotrophomonas maltophilia biofilms were established and confirmed by scanning electron microscopy and fluorescence microscopy with silver staining. The minimal inhibitory concentrations and biofilm inhibitory concentrations of commonly used antibiotics were determined.
Results47 of 51 strains were resistant to three or more antibiotics. 42 of 51 strains formed Stenotrophomonas maltophilia biofilms in vitro. Stenotrophomonas maltophilia biofilm formation greatly reduced sensitivity to most tested antibiotics, but not to levofloxacin. However, in the presence of erythromycin scanning electron microscopy revealed that levofloxacin inhibited Stenotrophomonas maltophilia biofilm formation. Factorial ANOVA revealed that erythromycin enhanced susceptibility to levofloxacin, cefoperazone/sulbactam, and piperacillin (p<0.05), and an ΔE model revealed that levofloxacin and erythromycin acted synergistically in biofilms, suggesting specific use of combined macrolide therapy may represent an effective treatment for Stenotrophomonas maltophilia infection.
ConclusionsAntibiotics could act synergistically to combat the protection conferred to clinical isolates of Stenotrophomonas maltophilia by biofilms. Macrolide antibiotics may be effective where used in combination.
Stenotrophomonas maltophilia (SMA) is an environmental pathogen and opportunistic Gram-negative bacterium that can infect immunocompromised patients or otherwise healthy patients when introduced by contaminated invasive medical devices.1 Dialysis technology, intubation, artificial implants and other widely employed medical materials can be colonized by bacteria, and SMA has been observed to form bacterial biofilms (BBF) on this equipment. In surgical departments, device-related contamination by potentially pathogenic bacteria can serve as a source for cross-infection,2 and nosocomial SMA infections have received increased attention in recent years.3–11 SMA bacteremia has been associated with mortality rates ranging from 14 to 69% in immunocompromised patients.12–14
Treatment of SMA infection is complicated by its natural resistance to many antimicrobial drugs, including carbapenems, and the rapid adaptation to the pulmonary environment.15 SMA can form BBF on host tissues, dramatically enhancing the resistance of SMA to therapeutically important antibiotics including aminoglycosides, fluoroquinolones, and tetracycline.16–20 Thus, biofilm formation represents an important mechanism of bacterial antibiotic resistance, and presents unique challenges in surgical medicine, complicating therapeutic management of such BBF.21,22
SMA biofilm formation was previously reported to be associated with resistance to ceftazidime, cefepime, ticarcillin/clavulanic acid, piperacillin/tazobactam, aztreonam, and gentamicin, but not to ciprofloxacin, levofloxacin, trimethoprim/sulfamethoxazole (TMP/SMX), or meropenem.23 The fluoroquinolone moxifloxacin was reported to interfere with SMA BBF formation24,25; however antibiotic resistance of clinical isolates has also been widely reported,26 mostly involving the study of strains isolated from cystic fibrosis (CF) patients.
In this study we sought to investigate the antibiotic-susceptibility of SMA strains isolated from invasive infections in non-CF patients. Using a methodology previously reported27,28 we established an in vitro model of SMA BBF, and investigated the antibiotic-susceptibility of SMA biofilms and planktonic bacteria. We assessed the capacity of antibiotics, applied individually and in combination, to reduce growth and biofilm formation of clinical isolates of SMA, in order to guide future clinical treatment of these patients.
Materials and methodsAntibiotic susceptibility of SMA isolatesClinical SMA strains were obtained from hospitalized patients with invasive infections that had originated from medical manipulation in the surgical wards and surgical ICUs of six general hospitals in Tianjin, China, between 2006 and 2012 (Table 1). The MICs of SMA to 12 antibiotics commonly used for Gram-negative bacilli were determined by microbroth dilution, analyzed according to the American National Clinical and Laboratory Standards Institute (CLSI) guidelines.29 The following strains were assessed in parallel for quality control: ATCC27853, ATCC25922 and ATCC25923, preserved in the Infectious Disease Institute of the Second Hospital of Tianjin Medical University, China.
In vitro model of SMA BBFUsing a methodology previously reported by Ceri27,28 we established an in vitro model of SMA BBF, in a Mueller–Hinton broth (MHB)-silica film system, as previously described.30,31 Cryopreserved SMA was recovered in sheep blood agar plates incubated aerobically overnight. A fresh single colony was transferred to fresh MHB and incubated for 8h at 35°C, from which a 200μL suspension of 0.5 McFarland was prepared and transferred to a 12-well flat-bottom plate, in which sterile silica film (1cm×1cm×1mm, L×W×T) and 1.8mL MHB were co-cultured at 35°C for 12 and 24h. After washing three times with 0.9% sodium chloride to remove planktonic bacteria, the BBF on the silica films was prepared. The culture medium was regarded as the negative control. Morphology was observed by scanning electron microscopy (SEM) and fluorescence microscopy (FSM) as described below.
Biofilm formation assessed using fluorescence microscopy with silver stainingAs previously described,32,33 the biofilm was fixed in 2.5% (v/v) glutaraldehyde in PBS (0.1M, pH 7.4) for 24h, then immersed in saturated calcium chloride solution for 15min, and rinsed with ddH2O between each step. The film was immersed in 5% silver nitrate solution for 15min, immediately stained with 1% hydroquinone for 2min, then rinsed with ddH2O. The film was fixed in 5% sodium thiosulfate solution for 2min, then rinsed in ddH2O and analyzed by FSM.
Biofilm formation assessed using scanning electron microscopyAs previously described,25 the silica biofilm was fixed in 2.5% (v/v) glutaraldehyde in PBS (0.1M, pH 7.4) at 4°C for 2h, fixed again with 1% osmic acid for 1h, then rinsed with PBS, dehydrated through a series of ethanol dilutions, then treated with isoamyl acetate. The specimen was dried in a vacuum, then coated with platinum–palladium and analyzed by SEM at 5–10kV.
Determination of the MICMIC values of the following antibiotics were calculated with a broth microdilution assay using the twofold dilution method according to the CLSI guidelines34: ciprofloxacin (CIP), levofloxacin (LEV), piperacillin (PIP), ceftazidime (CAZ), cefoperazone/sulbactam (SCF), erythromycin (ERY), sulfamethoxazole (SXT), and gentamycin (GM) within the range of CIP was 256–0.125mg/L, or 512–0.125mg/L.
Determination of the BICAfter dilution of the fresh SMA to 3×1010CFU/mL, 100μL of the culture was transferred to each well of a flat-bottom 96-well microtiter plate. As previously described,27,28,35,36 BBF were formed by immersing the pegs of a modified polystyrene microtiter lid into this plate, which was then incubated at 35°C for 24h. Peg lids were removed, rinsed in PBS, then placed on flat-bottom microtiter plates containing antibiotics, and incubated at 35°C. After 24h peg lids were removed, rinsed three times in PBS, then transferred onto flat-bottom microtiter plates containing 100μL of MHB per well, and biofilms were transferred from pegs to wells by centrifugation at 805×g for 20min. The optical density was measured at 650nm (OD650) on a microtiter plate colorimeter before and after incubation at 35°C for 6h.
Adequate biofilm growth was defined as a mean OD650 difference (OD650 at 6h minus the OD650 at 0h) of ≥0.05. BICs are defined as the lowest concentration of antibiotic in which the OD650 was 10% or less of the mean of the positive control well readings, representing at least a 1−log10 growth difference.
BBF combination sensitivity test by checkerboard methodThe susceptibility of biofilms to LEV, SCF and PIP alone and in combination with ERY was determined by the checkerboard method. Five strains of SMA (numbered 0020, 0037, 0040, 0088, 0256) were selected for the following test. Silica films were transferred to a 24-well flat-bottom microtiter plate. Then 2.0mL MHB was transferred to the well (blank control), 1.8mL MHB was transferred to each of the other wells. According to the MIC values of LEV, SCF and PIP against each strain, LEV, SCF and PIP was added to yield a final concentration of 1/2 MIC, 1 MIC and 2 MIC, and ERY was added to yield a final concentration of 1/16 MIC and 1/4 MIC. The plate was incubated at 35°C for 24h, then after washing with sterile physiological saline, the silica films were ultrasonicated at 60W for at 15min. The colony concentration was calculated from the OD650 by the ATCC27853 standard curve (Y=1.26514×10−8X+0.48028, with a linear range between 1.5×108 and 9.6×103CFU/mL, p<0.01). The density of colonies on the silica film was recorded from three wells in parallel.
Statistical analysisCFU per centimeter was indicated by mean±standard deviation, and the interaction of combined testing was analyzed by factorial experiment design ANOVA. The impact of antibiotics on biofilm growth was calculated using an ΔE model is based on the Bliss independence theory, described by the equation Ii=(IA+IB)−(IA×IB), where Ii is the predicted growth inhibition caused by the theoretical combination of drugs A and B, and IA and IB represent the growth inhibition caused by each drug individually. Since I=1−E, where E represents growth inhibition, the following equation is derived: Ei=EA×EB. Ei represented the predicted growth inhibition of the theoretical non-interactive combination of the drugs A and B, and EA and EB represent the growth inhibition caused by each drug individually. Interaction was described as the difference (ΔE) between the predicted and measured growth inhibition (ΔE=Epredicted−Emeasured). Statistically significant interactions of <100% were considered weak, those of 100–200% were considered moderate, and those >200% were considered strong.
ResultsAntibiotic susceptibility of clinical SMA isolatesThe antibiotic susceptibility of SMA isolates was assessed and in total, 47 (92.1%) of isolated strains were resistant to three or more antibiotics. The majority of tested strains were resistant to cefotaxime (94.1%) amikacin (90.2%) cefritaxone (88.2%) GM (82.4%) and CAZ (60.8%), and almost half of all tested strains were resistant to cefoperazone (49.0%), PIP (45.1%) and cefepime (45.1%).
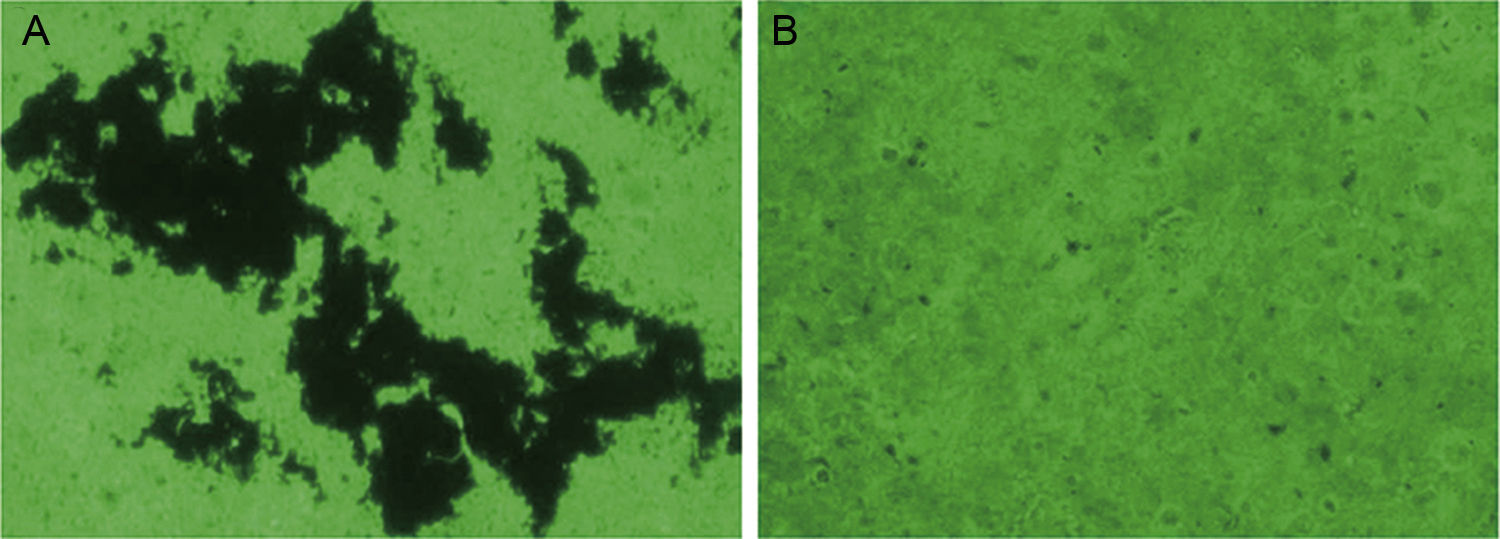
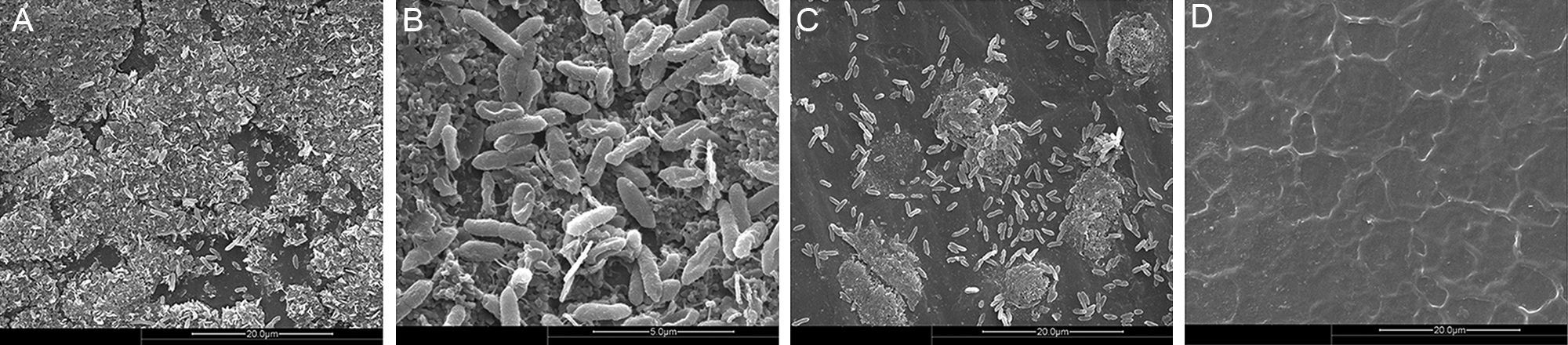
Clinical SMA isolates form BBFThe capacity of clinical isolates of SMA strain 0314 to form biofilms on the surface of the silica films incubated in MHB was assessed by fluorescent microscopy. Mature biofilms were formed by 42 of the 51 SMA isolates (82.35%). After 24h, silver staining revealed mature biofilm to be composed of black irregular sheets, scattered dots and black cottony membranes, and fine rod-shaped bacteria were observed at the borders (Fig. 1). No black aggregation was observed on the surface of silica film cultured in MHB in the absence of SMA. These findings were confirmed by SEM (Fig. 2). Biofilm ultrastructure was observed after 12h (Fig. 2C), and mature biofilm was observed at 24h (Fig. 2A and B). SMA was observed to be clustered on the surface of the biofilm, and SMA was mostly short rods of about 1.5μm×1.0μm, partially or entirely encompassed by the extracellular matrix (Fig. 2A and C). Fine bacilli alternated cross lots of extracellular mucus filaments, some cells were visible in split phase or undergoing apoptosis, and the bacterial community present on the biofilm was heterogeneous (Fig. 2B). No bacteria were observed on the surface of silica film cultured in MHB in the absence of SMA (Fig. 2D).
In vitro SMA BBF formation was visualized by fluorescence microscopy (×200). After 24h SMA strain 0314 cultured in MHB formed BBF on silica film. Black irregular sheets, scattered black dots, and cotton-like membranes were observed, and fine rod-shaped bacteria were observed at the borders (A). No black aggregation was observed on the surface of silica film cultured in MHB in the absence of SMA (B).
Ultrastructure of in vitro SMA BBF visualized by scanning electron microscopy. After 12h the biofilm of SMA strain 0314 cultured in MHB on silica film was observed (C), and was mature at 24h (A, B). SMA biofilm ultrastructure included clusters of proliferating bacteria (A, C). SMA was mostly short rods of about 1.5μm×1.0μm, partially or entirely encompassed by the extracellular matrix. A large number of fine bacilli alternated across numerous filamentous strands (B). No bacteria were observed on the surface of silica film cultured in MHB in the absence of SMA (D). B ×20,000, Bar=50μm; A, C, D ×5000; Bar=20μm.
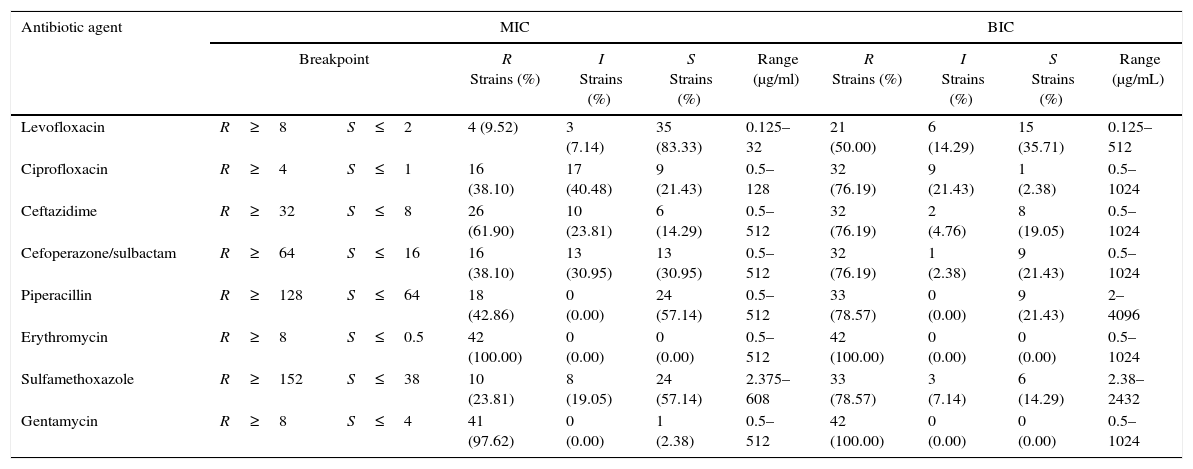
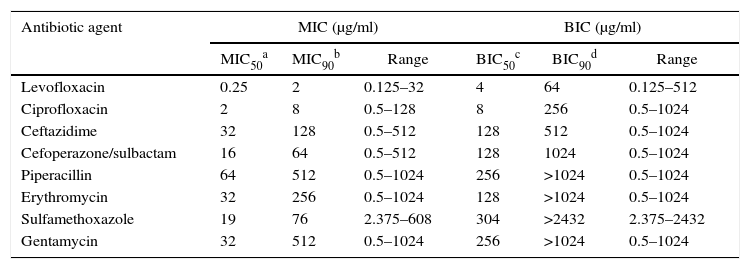
The effect of antibiotic agents on biofilm formation was analyzed by comparing MICs with BIC. The formation of biofilms enhanced the resistance of some strains to all tested antibiotics aside from GM and ERY, to which all the tested strains were resistant even in the absence of biofilm, and LEV (Table 2). After biofilm formation, 21 (50.0%) tested isolates were resistant to LEV, 33 (78.57%) to SXT, 33 (78.57%) to PIP, 32 (76.19%) to CAZ, 32 (76.19%) to SCF, and 32 (76.19%) to CIP. The average BIC90 was also much higher than the MIC90 (Table 3).
The Susceptibility of 42 SMA strains and their biofilms to 8 antibiotic agents.
| Antibiotic agent | MIC | BIC | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Breakpoint | R Strains (%) | I Strains (%) | S Strains (%) | Range (μg/ml) | R Strains (%) | I Strains (%) | S Strains (%) | Range (μg/mL) | ||
| Levofloxacin | R≥8 | S≤2 | 4 (9.52) | 3 (7.14) | 35 (83.33) | 0.125–32 | 21 (50.00) | 6 (14.29) | 15 (35.71) | 0.125–512 |
| Ciprofloxacin | R≥4 | S≤1 | 16 (38.10) | 17 (40.48) | 9 (21.43) | 0.5–128 | 32 (76.19) | 9 (21.43) | 1 (2.38) | 0.5–1024 |
| Ceftazidime | R≥32 | S≤8 | 26 (61.90) | 10 (23.81) | 6 (14.29) | 0.5–512 | 32 (76.19) | 2 (4.76) | 8 (19.05) | 0.5–1024 |
| Cefoperazone/sulbactam | R≥64 | S≤16 | 16 (38.10) | 13 (30.95) | 13 (30.95) | 0.5–512 | 32 (76.19) | 1 (2.38) | 9 (21.43) | 0.5–1024 |
| Piperacillin | R≥128 | S≤64 | 18 (42.86) | 0 (0.00) | 24 (57.14) | 0.5–512 | 33 (78.57) | 0 (0.00) | 9 (21.43) | 2–4096 |
| Erythromycin | R≥8 | S≤0.5 | 42 (100.00) | 0 (0.00) | 0 (0.00) | 0.5–512 | 42 (100.00) | 0 (0.00) | 0 (0.00) | 0.5–1024 |
| Sulfamethoxazole | R≥152 | S≤38 | 10 (23.81) | 8 (19.05) | 24 (57.14) | 2.375–608 | 33 (78.57) | 3 (7.14) | 6 (14.29) | 2.38–2432 |
| Gentamycin | R≥8 | S≤4 | 41 (97.62) | 0 (0.00) | 1 (2.38) | 0.5–512 | 42 (100.00) | 0 (0.00) | 0 (0.00) | 0.5–1024 |
R, resistant; I, intermediate susceptibility; S, susceptible.
Susceptibility of the bacteria in BBF was estimated from criteria set for planktonic cells, because of no identified criteria that were especially applicable to BBF. MICs, were determined by the microbroth two-fold dilution method. BICs, refer to a previously developed technique (Hill et al.,35 Moskowitz et al.,36 Olson et al.,27 Tomlin et al.28). The results represent all 42 isolates that could form mature biofilms in vitro in all 51 clinical strains.
Comparison of antibiotic susceptibility of the planktonic bacteria and biofilms of 42 SMA strains.
| Antibiotic agent | MIC (μg/ml) | BIC (μg/ml) | ||||
|---|---|---|---|---|---|---|
| MIC50a | MIC90b | Range | BIC50c | BIC90d | Range | |
| Levofloxacin | 0.25 | 2 | 0.125–32 | 4 | 64 | 0.125–512 |
| Ciprofloxacin | 2 | 8 | 0.5–128 | 8 | 256 | 0.5–1024 |
| Ceftazidime | 32 | 128 | 0.5–512 | 128 | 512 | 0.5–1024 |
| Cefoperazone/sulbactam | 16 | 64 | 0.5–512 | 128 | 1024 | 0.5–1024 |
| Piperacillin | 64 | 512 | 0.5–1024 | 256 | >1024 | 0.5–1024 |
| Erythromycin | 32 | 256 | 0.5–1024 | 128 | >1024 | 0.5–1024 |
| Sulfamethoxazole | 19 | 76 | 2.375–608 | 304 | >2432 | 2.375–2432 |
| Gentamycin | 32 | 512 | 0.5–1024 | 256 | >1024 | 0.5–1024 |
The effects of combining ERY with other antibiotics was investigated in five SMA isolates which were determined to have moderate susceptibility to LEV, SCF and PIP. The MIC of LEV in the chosen strains (0020, 0037, 0040, 0080 and 0256) was between 0.5 and 64mg/L, the MIC of SCF was between 8 and 32mg/L and the MIC of PIP was between 2 and 64mg/L. Addition of ERY did not significantly enhance the efficacy of SCF (p=0.06), but did significantly enhance LEV and PIP efficacy (all p<0.05). Factorial ANOVA analysis revealed that the efficacy of SCF, LEV, and PIP was significantly improved in the presence of ERY (F=8.460, p=0.000; F=20.825, p=0.000; and F=4.506, p=0.001, respectively; Table 4, and one-way ANOVA, p<0.05). The synergy of antibiotics was assessed by the ΔE model, and the combination of ERY and LEV was found to have a synergistic effect against four of the five strains tested (Table 5).
Effect of antibiotics alone and in combination on the SMA biofilms.
| Antibiotic agent | Colony density (104CFU/cm2)a | ||||
|---|---|---|---|---|---|
| – | ½ MIC | 1 MIC | 2 MIC | ||
| SCF | No ERY | 149.6±3.8 | 150.80±3.7 | 153.40±4.9 | 152.20±4.8 |
| ERY 1/16 MIC | 149.2±5.0 | 147.60±6.3 | 140.00±3.4 | 132.80±4.7 | |
| ERY ¼ MIC | 150.2±4.0 | 141.00±5.4 | 132.20±3.3 | 130.00±4.5 | |
| PIP | No ERY | 148.4±5.6 | 146.20±3.4 | 153.20±3.7 | 148.20±5.5 |
| ERY 1/16MIC | 147.4±4.8 | 144.60±5.5 | 143.80±5.4 | 133.20±5.2 | |
| ERY 1/4MIC | 150.6±5.8 | 143.40±5.9 | 140.40±4.6 | 130.80±3.7 | |
| LEV | No ERY | 153.0±4.1 | 131.80±4.5 | 117.40±5.1 | 108.20±4.9 |
| ERY 1/16MIC | 149.2±5.4 | 120.40±3.6 | 89.80±4.4 | 80.60±3.9 | |
| ERY 1/4MIC | 151.2±4.3 | 96.00±7.5 | 75.60±5.8 | 66.20±5.5 | |
LEV, levofloxacin; PIP, piperacillin; SCF, cefoperazone/sulbactam; ERY, erythromycin.
Combination tests all revealed a statistical interaction by factorial experiment design and ANOVA, of SCF with ERY (F=8.460, p=0.000), LEV with ERY (F=20.825, p=0.000), and PIP with ERY (F=4.506, p=0.00) respectively.
In vitro interactions of ERY in combination with SCF, PIP, and LEV against BBF, and as indicated by the ΔE model.
| Strains | E value mean (range) | ∑Syn (n)%a | ∑Ant (n)%b | Interpretation | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SCF | PIP | LEV | SCF | PIP | LEV | SCF | PIP | LEV | SCF | PIP | LEV | |
| 0020 | 8.97 (−0.64 to 16.62) | 9.43 (2.05 to 15.78) | 17.38 (−0.85 to 29.65) | 54.44 (5) | 56.55 (6) | 105.13 (5) | −0.64 (1) | 0 (0) | −0.85 (1) | NI | NI | SYN |
| 0037 | 15.37 (4.13 to 22.26) | 4.18 (−2.04 to 8.96) | 27.29 (12.17 to 38.71) | 92.32 (6) | 27.09 (5) | 163.72 (6) | 0 (0) | −2.04 (1) | 0 (0) | NI | NI | SYN |
| 0040 | 10.05 (1.34 to 18.15) | 3.54 (−10.83 to 15.75) | 19.33 (8.55 to 28.94) | 60.29 (6) | 34.88 (4) | 115.96 (6) | 0 (0) | −13.62 (2) | 0 (0) | NI | NI | SYN |
| 0088 | 7.64 (−0.68 to 13.68) | 7.10 (2.04 to 13.33) | 12.77 (0.10 to 20.21) | 46.10 (6) | 42.57 (60 | 76.63 (6) | 0 (0) | 0 (0) | 0 (0) | NI | NI | NI |
| 0256 | 9.01 (5.39 to 12.84) | 12.57 (8.06 to 17.99) | 17.37 (6.12 to 25.99) | 54.06 (6) | 75.44 (6) | 104.21 (6) | 0 (0) | 0 (0) | 0 (0) | NI | NI | SYN |
NI, no interaction; SYN, synergism; ANT, antagonism; n, number of interactions.
a and b were the sums of the percentages of all statistically significant synergistic and antagonistic interactions.
SMA strain 0020 biofilm formation was also assessed in the presence of SCF, LEV, PIP and/or ERY (Fig. 3). We observed that LEV damaged BBF in the presence or absence of ERY (Fig. 3A and B), but the combination of ERY with LEV reduced the number of bacteria anchored to the BBF, thinned the polysaccharide matrix and altered bacterial morphology. Some spherical, irregularly shaped, or cracked bacteria were observed. In the absence of antibiotics, the BBF was mature and bacterial cells were packaged in a lot of sticky polysaccharide matrix. Bacterial growth was strong and some bacteria were observed to be in the division phase (Fig. 3C). Neither PIP nor SCF, in the presence or absence of ERY, altered the state of the bacteria or biofilm (Fig. 3D).
Impact of antibiotic agents on ultrastructure of in vitro BBF visualized by scanning electron microscopy. SMA strain 0020 BBF formed a complete mature structure in which the bacterial were packaged with dense matrix (C). In the presence of erythromycin (B) or erythromycin and levofloxacin (A) the BBF matrix became thin, fewer adhered bacteria were observed and some exhibited spherical or irregular form. The black arrow indicates a bacterial cell undergoing division phase. In the presence of erythromycin and piperacillin the BBF remained similar to that seen in the blank control (D). D ×20,000; Bar=100μm; A, B, C ×5000; Bar=50μm.
Treatment of nosocomial SMA infections is complicated by high rates of antibiotic resistance. Although pharmacokinetics and drug penetration influence the clinical efficacy of antibiotics against planktonic bacterial, resistance can be characterized in in vitro susceptibility tests. In this study we sought to investigate the antibiotic-susceptibility of SMA strains isolated from invasive infections in non-CF patients. We established an in vitro model of SMA BBF, and investigated the antibiotic-susceptibility of SMA biofilms and planktonic bacteria. Fifty-one clinical isolates of SMA were taken from patients infected with SMA through medical manipulation and/or immunosuppressive therapy in the surgical departments of Tianjin hospitals.
In our sample the vast majority of isolated strains (92.16%) were resistant to three or more antibiotics. The highest rate of resistance was to cefotaxime (94.1%) and the lowest (only 13.7%) was to LEV. The intrinsic or acquired resistance of SMA to antibiotics has been previously reported, and drastically reduces the antibiotic options available for treatment.22
BBF formation was also reported to enhance antibiotic resistance. Bacterial adhesion involves development of pili, changes in cell surface and hydrophobicity, and is crucial to BBF formation,37,38 but the mechanism of BBF formation varies by attachment substrate.38 We selected silicon film as an attachment matrix because of its superior resistance to corrosion, chemical stability and non-tackiness. A majority of SMA strains included in our study formed biofilms in vitro, and mature biofilms were observed within 24h, suggesting that transmission of these clinical isolates may have been facilitated by development of biofilms on clinical tools. BBF structure was identified by silver staining characterized by SEM ultrastructure analysis. The extracellular matrix around the bacteria appeared to be made up of secreted exopolysaccharide. BBF formation was previously reported to be associated with S. maltophilia fimbriae 1 (SMF-1), which is composed of fimbrin subunits and shares significant similarity with the N-terminal amino acid sequences of several fimbrial adhesins of pathogenic Escherichia coli strains and the Cup A fimbriae in Pseudomonas aeruginosa.39
In our sample, all clinical isolates were more resistant to antibiotics in the BBF than in planktonic state. BBF have been previously reported to confer antibiotic resistance of up to 1000-fold,16–20 and thus BBF formation is thought to be an important mechanism of antibiotic resistance by SMA. The BBF constitute a positively charged polysaccharide matrix barrier to antibiotics with a negative charge, such as aminoglycosides.21 The extracellular polysaccharide matrix can also absorb high levels of extracellular enzymes, such as β-lactamase. We found that most isolates were resistant to β-lactams, including third and fourth generation cephalosporins, although resistance to cefoperazone/sulbactam was lower. SMA strains wrapped in extracellular polysaccharide matrices are less able to obtain molecular oxygen and nutrients, and to eliminate metabolites, which can cause bacterial dormancy or retarded growth rates, and local metabolite accumulation.22 Thus, in this study, antimicrobial agents that target bacterial growth, such as β-lactams, aminoglycosides, and LEV were less effective. Due to local metabolite accumulation, the microenvironment of bacteria living deep in tissues is acidic, which inactivates most of the therapeutic drugs that optimally act in neutral conditions.
SMA gene expression profiles have been reported to change significantly on BBF formation, presenting a range of mechanisms for development of antibiotic resistance.40
Development of BBF also enables evasion of the host immune response. The glycocalyx shell protects bacteria from phagocytosis and secreted quorum-sensing factor N-(3-oxododecanoyl)-l-homoserine lactone (HSL) and C4-HSL can inhibit leukocyte proliferation and cytokine secretion.41 Protection from the host immune response provides a protected niche in which SMA can evolve resistance to previously effective antimicrobials.
In this study, formation of biofilm conferred resistance to all tested antimicrobials aside from LEV, according to CLSI break points. LEV alone inhibits mature BBF in a concentration-dependent manner, which indicates that the in vivo maximum permissible dosage for BBF should be investigated. Complete elimination of biofilm infection remains challenging, but inhibiting bacterial adherence or destroying the matrix may prove useful tactics, in combination with activated host immunity. Macrolides have been reported to assist other antibiotics in bactericidal effects on P. aeruginosa BBF, by inhibiting synthesis of GDP-mannose dehydrogenase, the main component of biofilms.42,43 Moreover, macrolides can inhibit neutrophil accumulation, and promote improved CD4+/CD8+ T cell ratios.44,45 In this study ERY, a 14-membered ring macrolide, acted synergistically with LEV, SCF and PIP, even where SMA strains were resistant to ERY alone. ERY markedly enhanced efficacy of LEV and was observed to reduce bacterial adhesion to biofilms and undermined biofilm architecture. We selected LEV, SCF and PIP to test in combination with ERY based on their BIC value, but other combinations of antimicrobials may also show clinical effectiveness. Therefore, further combinations of antimicrobials should be tested in future.
In summary, we found that antibiotics could act synergistically to combat the protection conferred to clinical isolates of SMA by BBF. Our findings suggest that macrolide antibiotics may be effective where used in combination. However further specific in vivo studies will be required to confirm whether this approach can treat nosocomial SMA infection in the clinic.
FundingThe study was supported by the National Natural Science Foundation of China (Grant No.: 81402095).
Conflicts of interestThe authors declare no conflicts of interest.