The aim of this study was to compare the predictions of Framingham cardiovascular (CV) risk score (FRS) and the American College of Cardiology/American Heart Association (ACC/AHA) risk score in an HIV outpatient clinic in the city of Vitoria, Espirito Santo, Brazil. In a cross-sectional study 341 HIV infected patients over 40 years old consecutively recruited were interviewed. Cohen's kappa coefficient was used to assess agreement between the two algorithms. 61.3% were stratified as low risk by Framingham score, compared with 54% by ACC/AHA score (Spearman correlation 0.845; p<0.000). Only 26.1% were classified as cardiovascular high risk by Framingham compared to 46% by ACC/AHA score (Kappa=0.745; p<0.039). Only one out of eight patients had cardiovascular high risk by Framingham at the time of a myocardial infarction event registered up to five years before the study period. Both cardiovascular risk scores but especially Framingham underestimated high-risk patients in this HIV-infected population.
The World Health Organization (WHO) estimates that the number of AIDS related deaths reached about 1.1 million at the end of 2015.1 The overall incidence of AIDS related deaths due to opportunistic infections and AIDS defining cancers has decreased significantly with widespread availability of effective antiretroviral treatment.2 People living with HIV are living long enough to experience non-AIDS defining illnesses. These events also called chronic noncommunicable diseases include diabetes, chronic obstructive pulmonary disease, kidney diseases, hypertension, cardiovascular diseases, and cancer.3
Cardiovascular disease (CVD) is one of the most important causes of mortality among adults in developed countries. Furthermore, among HIV-infected patients the estimated CVD risk is 61% greater compared with uninfected controls, according to a meta-analysis,4 and the risk for sudden cardiac death is about 4-fold greater.5 Immune dysfunction and persistent inflammation features of HIV infection have a possible role in the pathogenesis of CVD risk in addition to traditional risk factors such as cigarette smoking, elevated blood pressure and total cholesterol. Although findings from the SMART study clearly demonstrated fewer CVD events in patients treated with ART compared to those interrupting therapy,6 large prospective cohort studies as D.A.D study have shown excess CVD risk associated with some specific ARV drugs.7,8
WHO recommends that assessment and management of cardiovascular risk should be provided for all individuals living with HIV according to standard protocols used for the general population.9 Brazilian HIV treatment guidelines recommend screening all HIV adult patients for cardiovascular risk using Framingham score risk, before therapy and at least yearly.10 However, the best algorithm for predicting CVD risk remains controversial. The American College of Cardiology and American Heart Association (ACC/AHA) developed their own assessment of cardiovascular risk.11 As none of those risk scores has been validated on HIV populations, it seems important to assess how these two scores agree in real life.
MethodsThis was a cross-sectional analysis performed at an AIDS outpatient clinic from January 2015 to July 2015. This clinic is one of the most important specialized referral services (SAS) in the city of Vitoria, in southeast Brazil, and is part of the national public network providing care for HIV-infected patients in the country. All HIV-infected patients over 40 years old were invited to participate and those who agreed were selected, after providing their written informed consent.
Individual information was collected either by the examiner during the interview, or abstracted from the patients’ medical records if obtained in the last three months. Data included age, sex, race, years since HIV diagnosis, probable means of transmission, last HIV-1 viral load and T CD4+ lymphocytes count, systolic blood pressure measurement, total cholesterol and high density lipoprotein cholesterol (HDL) levels, antiretroviral therapy (ARV) used, anti-hypertensive therapy, and associated conditions like cigarette smoking, diabetes mellitus, systemic arterial hypertension (SAH), and previous myocardial infarction. The few patients with previous myocardial infarction had their cardiovascular score risks measured using information of the medical records at the time the event had occurred. These patients as well as those with diabetes were further stratified as high risk. The Framingham and ACC/AHA scores for all other patients were calculated according to algorithms accessed respectively at http://my.americanheart.org/cvriskcalculator and at http://www.cardiosourse.org/science-and-quality/practice-guidelines-and-quality-standards/2013-prevention-guideline-tools.aspx. IBM SPSS statistics version 23 (SPSS, Chicago, IL) was used for statistical analysis. Continuous variables are presented as medians and interquartile range and categorical variables as percentages. Spearman correlation was used to compare values of Framingham and ACC/AHA scores and Kappa coefficient was used to assess agreement beyond chance between these two scores. The goal of this study was to apply and to compare different kinds of scores for CVD risk in an HIV-infected population. The study was conducted after approval by the Institution Ethical Committee for Research. All participants gave their written informed consent.
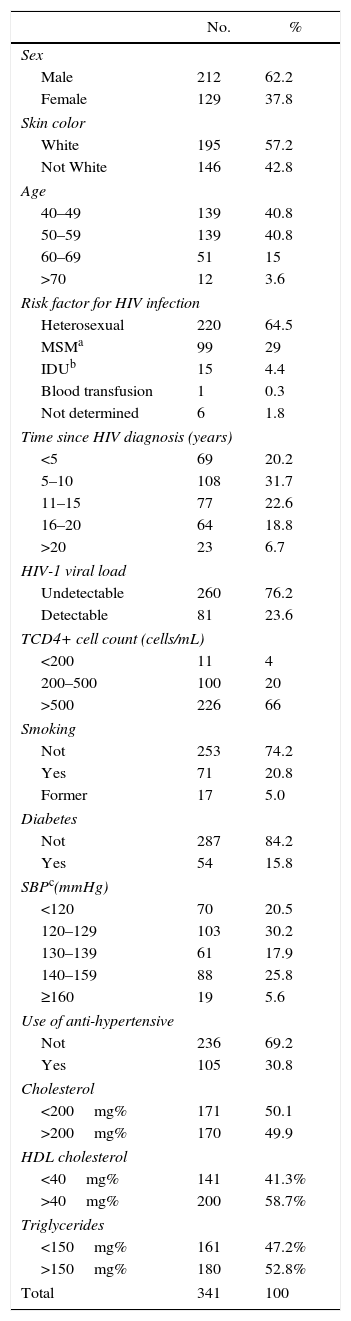
ResultsA total of 341 HIV-infected patients were included in the analysis. Table 1 presents baseline characteristics for the included patients. Most of them (62.2%) were male; 57.2% white, median age at enrollment 51 (IQR 46–57) years.
Characteristics of HIV-infected patients on treatment at Santa Casa Misericórdia, Vitória, ES, Brazil, 2015.
| No. | % | |
|---|---|---|
| Sex | ||
| Male | 212 | 62.2 |
| Female | 129 | 37.8 |
| Skin color | ||
| White | 195 | 57.2 |
| Not White | 146 | 42.8 |
| Age | ||
| 40–49 | 139 | 40.8 |
| 50–59 | 139 | 40.8 |
| 60–69 | 51 | 15 |
| >70 | 12 | 3.6 |
| Risk factor for HIV infection | ||
| Heterosexual | 220 | 64.5 |
| MSMa | 99 | 29 |
| IDUb | 15 | 4.4 |
| Blood transfusion | 1 | 0.3 |
| Not determined | 6 | 1.8 |
| Time since HIV diagnosis (years) | ||
| <5 | 69 | 20.2 |
| 5–10 | 108 | 31.7 |
| 11–15 | 77 | 22.6 |
| 16–20 | 64 | 18.8 |
| >20 | 23 | 6.7 |
| HIV-1 viral load | ||
| Undetectable | 260 | 76.2 |
| Detectable | 81 | 23.6 |
| TCD4+ cell count (cells/mL) | ||
| <200 | 11 | 4 |
| 200–500 | 100 | 20 |
| >500 | 226 | 66 |
| Smoking | ||
| Not | 253 | 74.2 |
| Yes | 71 | 20.8 |
| Former | 17 | 5.0 |
| Diabetes | ||
| Not | 287 | 84.2 |
| Yes | 54 | 15.8 |
| SBPc(mmHg) | ||
| <120 | 70 | 20.5 |
| 120–129 | 103 | 30.2 |
| 130–139 | 61 | 17.9 |
| 140–159 | 88 | 25.8 |
| ≥160 | 19 | 5.6 |
| Use of anti-hypertensive | ||
| Not | 236 | 69.2 |
| Yes | 105 | 30.8 |
| Cholesterol | ||
| <200mg% | 171 | 50.1 |
| >200mg% | 170 | 49.9 |
| HDL cholesterol | ||
| <40mg% | 141 | 41.3% |
| >40mg% | 200 | 58.7% |
| Triglycerides | ||
| <150mg% | 161 | 47.2% |
| >150mg% | 180 | 52.8% |
| Total | 341 | 100 |
According to patients’ self reports, unprotected heterosexual activity was the most frequent mode (64.5%) of HIV acquisition, while 29% reported homosexual sex and only 4.4% reported injecting drug use.
Patients who have been diagnosed with HIV in the last 5–10 years represented 31.7% of total, while 20.2% have been diagnosed recently (less the five years ago). The proportion of patients diagnosed in the last 11–15 years was 22.6%, similar to those who knew they have the virus for 16–20 years (18.8%). Only 6.7% had been diagnosed with HIV for more than 20 years. Almost all patients (97.9%) were on ART. The backbone of nucleoside transcriptase inhibitors of most patients used was TDF+3TC (54.3%) followed by AZT+3TC (37%). The third drug most commonly used was Efavirenz (39.9%) followed by Lopinavir/r (26.7%), Atazanavir/r (16.4%), and Nevirapine (7%). TCD4 cell count was above 500cells/mL in 66% of patients and HIV viral load was bellow detection limits in 76.2% of patients.
Diabetes was found in 15.8% of patients and 30.8% were on current treatment for SAH. Median SBP was 130mmHg (IQR 120–140). Cigarette smoking was reported by 20.8% while 5% declared to be former smokers. Median total cholesterol level was 199 (IQR 167–231)mg% and levels above 200mg% were detected in 49.9% of the patients. HDL levels below 40mg% was found in 41.3% of the patients, while median HDL level was 42 (IQR 36–50)mg%. Triglycerides were above 150mg% in 52.8% of the individuals, median level was 155 (IQR 105–229)mg%.
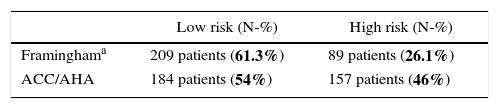
The assessment of cardiovascular risk by Framingham and ACC/AHA score risks are shown in Table 2. Among 209 patients classified as low risk by Framingham score, 184 were also classified as low risk by ACC/AHA score (Spearman correlation 0.845; p<0.000). Among 43 patients classified as intermediate risk by Framingham score, 37 were classified as high risk by ACC/AHA score. Therefore, lower concordance for high risk between the two scores was observed, as 26.1% were classified as high risk by Framingham and 46% by ACC/AHA score (Kappa=0.745; p<0.039).
Comparison between cardiovascular risk estimation using Framingham and ACC/AHA algorithms.
| Low risk (N-%) | High risk (N-%) | |
|---|---|---|
| Framinghama | 209 patients (61.3%) | 89 patients (26.1%) |
| ACC/AHA | 184 patients (54%) | 157 patients (46%) |
Acute myocardial infarction was related by nine patients up to five years before the study period, eight were male, eight were treating SAH, one had diabetes mellitus, and four were previous smokers. Efavirenz was used by four patients, Atazanavir/r by three patients, and Lopinavir/r by two patients. Median TCD4 was 540cells/mL and all but one of these patients had HIV-1 viral load below detection limits. Data for estimation of score risks were available from medical records at the time of the event for eight patients. Five out of eight patients had high score risk by ACC/AHA compared to only one by Framingham score.
DiscussionIn this cross-sectional analysis of an HIV-infected cohort on treatment we observed an important prevalence of traditional cardiovascular risk factors such as diabetes (15.8%), cigarette smoking (20.8%), and SAH (30.8%). These rates are higher than those observed in the general population in Brazil, according to a regular phone call surveillance conducted by the Ministry of Health, respectively 8% for diabetes, 10.8% for cigarette smoking, and 24.8% for SAH.12 The average years of life lost due to smoking among HIV-infected patients was estimated as 12.3 years, which is more than twice that for the general population.13 In Brazil, the prevalence of smoking reduced in the last decades but remains high especially in cities in the south and southeast regions.14
In our study the agreement between Framingham and ACC/AHA scores was lower for identifying high-risk patients. Conflicting results have been reported comparing different cardiovascular risk scores among HIV-infected populations in different countries.15–19 Curiously, the Framingham risk score overestimated heart events in a Mediterranean HIV-infected population and a Regicor chart (an adaption for the characteristics of the Spanish population) worked better as a predictor for ischemic CV events.20 Some intrinsic characteristics of this Spanish population may explain this different impact of Framingham score as it usually underestimates CV risk in HIV-infected populations. Only 26.1% of our patients were stratified as high-risk by Framingham compared to 46% by ACC/AHA. Only one out of eight patients was categorized as high-risk according to Framingham score at the time of a myocardial infarction.
Dyslipidemia remains an important issue in our outpatient clinic. We have shown before that use of boosted protease inhibitors, especially lopinavir/r, was associated with high cholesterol levels.21 Friis-Moller et al. have demonstrated in the D:A:D study that the additional risk of CVD among patients receiving lopinavir/r ranged from 8 to 12% per year, almost doubling the risk over 10 years.22 Most patients on statin therapy were using a weak statin, pravastatin, that was not always available for all patients in need. Use of atorvastatin in HIV-infected patients on therapy was reported to reduce the total coronary artery plaque volume.23 The multicenter REPRIEVE trial aimed to assess the potential benefits of statin therapy (pitavastatin) in preventing CVD in people with HIV.24
Our study has important limitations. It is a cross-sectional analysis comparing two different CV risk scores. Certainly, the best way to compare those scores should be a prospective study observing how these scores work as an HIV cohort grows older. However, as patients with HIV live longer on therapy, the effect of CVD on morbidity and mortality will probably worsen unless effective management strategies are developed. Traditional screening methods for CVD might underestimate the risk of HIV-infected patients as these methods do not take into account non-traditional risk factors involved in the development of atherosclerosis.
Conflicts of interestThe authors declare no conflicts of interest.