This work performed a phenotypic and genotypic characterization of 79 clinical isolates of Enterobacteriaceae and Pseudomonadaceae collected in hospitals of Southern Ecuadorin 2013. Our results showed a high incidence of β-lactamases and ESBLs with blaTEM and blaCTX-M as the prevalent genes, respectively. By direct sequencing of PCR amplicons, the different β-lactamases and variants of the genes were also distinguished. Our results revealed a predominance of TEM-1 β-lactamase and the presence of different CTX-M variants with a prevalence of CTX-M-15. Two infrequent CTX-M variants in South America were also identified. To the best of our knowledge, this is one of the first studies describing the genetic characteristics of β-lactamases in Ecuador.
Since the discovery of antibiotics, many pathogens which had been considered defeated have continued to develop resistance to these drugs. The emergence of resistant microorganisms to antibiotics is a problem that has extended globally.1,2 This phenomenon is widespread in hospitals mainly because of the large and often inappropriate use of antimicrobials. However, due to the bacteria's ability to exchange genetic information, environmental and nosocomial pathogens have developed the same resistance mechanisms, since most resistance genes have their probable origin in environmental microorganisms.3 Resistant pathogens to most antibiotics have already been described, but in particular it is of great impact the resistance to β-lactams. Resistance to these antimicrobial agents is due to the production of β-lactamases, and the genes encoding these enzymes have been found worldwide mainly in Gram-negative bacteria, prevalently in Enterobacteriaceae and Pseudomonas spp.4,5
Among β-lactamases, extended-spectrum β-lactamases (ESBLs), AmpC-type β-lactamases, and carbapenemases are of great concern. In particular ESBLs are the most abundant in Enterobacteriaceae, with more than 600 natural variants (http://www.lahey.org/Studies/), and with a prevalence of CTX-M family that became predominant over TEM and SHV during the first decade of the 21st century.6
As far as we know, there is a lack of detailed reports characterizing the β-lactamase spread in Ecuador.7,8 In this work a phenotypic and molecular characterization of β-lactamases was performed in clinical pathogens isolated in two main public hospitals of the Loja province, Ecuador.
The clinical isolates of Enterobacteriaceae and Pseudomonas aeruginosa were obtained from patients of Ygnacio Monteros and IESS Hospitals. The samples were collected between May and November 2013, while the bacterial cultures were grown in agar-MacConkey Petri dishes, identified by traditional biochemical testing and validated by Microgen™ GnA+B-ID system.
Antimicrobial susceptibility testing was performed by Kirby-Bauer disk diffusion procedure according to CLSI guidelines.9 The isolates were tested for susceptibility to amikacin (AMK), amoxicillin/clavulanic acid (AMC), ampicillin (AMP), ampicillin/sulbactam (SAM), aztreonam (ATM), cefepime (FEP), cefotaxime (CTX), cefoxitin (FOX), ceftazidime (CAZ), ciprofloxacin (CIP), imipenem (IMP), meropenem (MEM), netilmicin (NET), and piperacillin/tazobactam (PTZ). The investigation of phenotypic production of ESBL and AmpC-type β-lactamase was performed by techniques of double disk synergism (DDST) on Mueller Hinton agar and combination disk method. In particular for ESBL detection, DDST was performed on agar with AMC-20/10μg disk positioned 25mm (center-to-center) away from disks containing CAZ-30μg, CTX-30μg, FEP-30μg and ATM-30μg, and the confirmation combination disk method with disks of CTX-30μg, cefotaxime/clavulanic acid (30/10μg), CAZ-30μg and ceftazidime/clavulanic acid (30/10μg)10; for AmpC-type β-lactamase detection, DDST (for constitutive AmpC) was performed with boronic acid containing disk (30μg) placed at a center-to-center distance to a CAZ-30μg and a CTX-30μg disk of 25mm, while for combination disk method (for inducible AmpC), disks of CTX-30μg, CAZ-30μg and ATM-30μg were placed close to a IMP-10μg.11,12
Three multiplex PCR and one simplex PCR were performed to determine the presence of blaTEM, blaSHV, blaOXA-1-like and blaCTX-M genes in ESBL-producing isolates, and blaACC, blaFOX, blaMOX, blaDHA, blaLAT, blaCMY, blaMIR and blaACT genes in plasmid-mediated AmpC β-lactamase-producing isolates. PCR primers and procedures were performed as previously described.13
Purification and sequencing of PCR products were carried out by Macrogen Inc. (Korea) and the types of β-lactamase genes were identified by comparison with the sequences in GenBank (http://www.ncbi.nlm.nih.gov/genbank/).
During the study, a total of 79 isolates resistant to β-lactams were recovered. The most frequent isolated bacterial species were Escherichia coli (39) followed by Klebsiella pneumoniae (9), Klebsiella oxytoca (7) Enterobacter aerogenes (7), Enterobacter cloacae (5) Proteus mirabilis (3), Pseudomonas aeruginosa (3), Citrobacter freundii (3), Proteus vulgaris (2), and Enterobacter koseri (1).
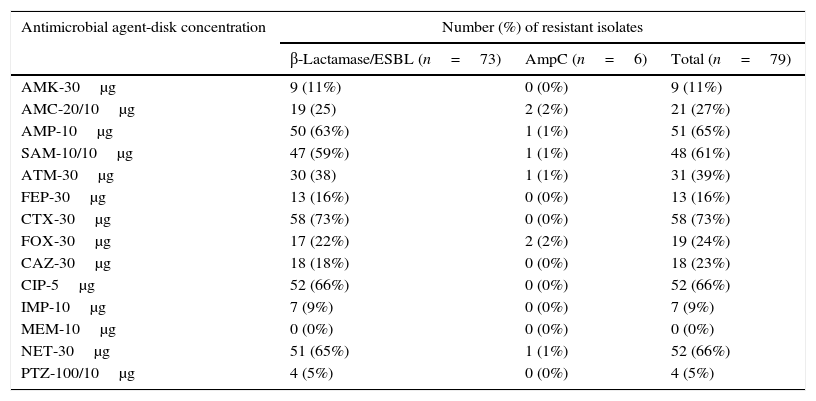
All isolates showed antimicrobial resistance to one or more β-lactam antibiotics, and among them, 73 were confirmed to be positive for β-lactamase/ESBL and six for AmpC β-lactamase production (see Table 1 for details).
Resistance rate for the clinical isolates.
| Antimicrobial agent-disk concentration | Number (%) of resistant isolates | ||
|---|---|---|---|
| β-Lactamase/ESBL (n=73) | AmpC (n=6) | Total (n=79) | |
| AMK-30μg | 9 (11%) | 0 (0%) | 9 (11%) |
| AMC-20/10μg | 19 (25) | 2 (2%) | 21 (27%) |
| AMP-10μg | 50 (63%) | 1 (1%) | 51 (65%) |
| SAM-10/10μg | 47 (59%) | 1 (1%) | 48 (61%) |
| ATM-30μg | 30 (38) | 1 (1%) | 31 (39%) |
| FEP-30μg | 13 (16%) | 0 (0%) | 13 (16%) |
| CTX-30μg | 58 (73%) | 0 (0%) | 58 (73%) |
| FOX-30μg | 17 (22%) | 2 (2%) | 19 (24%) |
| CAZ-30μg | 18 (18%) | 0 (0%) | 18 (23%) |
| CIP-5μg | 52 (66%) | 0 (0%) | 52 (66%) |
| IMP-10μg | 7 (9%) | 0 (0%) | 7 (9%) |
| MEM-10μg | 0 (0%) | 0 (0%) | 0 (0%) |
| NET-30μg | 51 (65%) | 1 (1%) | 52 (66%) |
| PTZ-100/10μg | 4 (5%) | 0 (0%) | 4 (5%) |
Abbreviations: AMK, amikacin; AMC, amoxicillin/clavulanic acid; AMP, ampicillin; SAM, ampicillin/sulbactam; ATM, aztreonam; FEP, cefepime; CTX, cefotaxime; FOX, cefoxitin; CAZ, ceftazidime; CIP, ciprofloxacin; IMP, imipenem; MEM, meropenem; NET, netilmicin; PTZ, piperacillin/tazobactam.
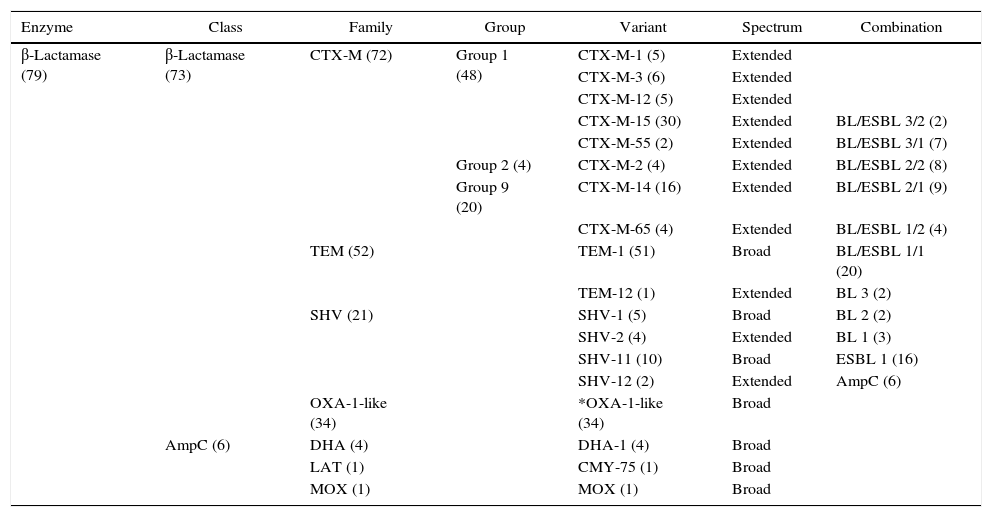
By PCR amplification, it was found that 31% of the samples showed the presence of one β-lactamase gene, whilst isolates often carried two (28%), three (20%), four (18%), and occasionally five (3%) different variants of β-lactamase enzymes (see Table 2 for details).
Summary of β-lactamase encoding genes detected in ESBL-producing bacteria from clinical isolates.
| Enzyme | Class | Family | Group | Variant | Spectrum | Combination |
|---|---|---|---|---|---|---|
| β-Lactamase (79) | β-Lactamase (73) | CTX-M (72) | Group 1 (48) | CTX-M-1 (5) | Extended | |
| CTX-M-3 (6) | Extended | |||||
| CTX-M-12 (5) | Extended | |||||
| CTX-M-15 (30) | Extended | BL/ESBL 3/2 (2) | ||||
| CTX-M-55 (2) | Extended | BL/ESBL 3/1 (7) | ||||
| Group 2 (4) | CTX-M-2 (4) | Extended | BL/ESBL 2/2 (8) | |||
| Group 9 (20) | CTX-M-14 (16) | Extended | BL/ESBL 2/1 (9) | |||
| CTX-M-65 (4) | Extended | BL/ESBL 1/2 (4) | ||||
| TEM (52) | TEM-1 (51) | Broad | BL/ESBL 1/1 (20) | |||
| TEM-12 (1) | Extended | BL 3 (2) | ||||
| SHV (21) | SHV-1 (5) | Broad | BL 2 (2) | |||
| SHV-2 (4) | Extended | BL 1 (3) | ||||
| SHV-11 (10) | Broad | ESBL 1 (16) | ||||
| SHV-12 (2) | Extended | AmpC (6) | ||||
| OXA-1-like (34) | *OXA-1-like (34) | Broad | ||||
| AmpC (6) | DHA (4) | DHA-1 (4) | Broad | |||
| LAT (1) | CMY-75 (1) | Broad | ||||
| MOX (1) | MOX (1) | Broad |
The numbers in brackets represent the number of isolates (in enzyme and class columns) and of found genes (in family, group, variant and combination columns). The last column represents the different combinations of β-lactamase (BL), ESBL and AmpC genes found. *In the case of blaOXA-1-like, it was not possible to distinguish the different variants by direct sequencing of PCR amplicons.13
The genes encoding CTX-M were the most common and were found in 63 of the 73 ESBL-producing isolates. In particular the genes blaCTX-M belonging to phylogenetic groups 1, 2 and 9 were found in 48, 4 and 20 isolates, respectively. It was also detected that 52 of the ESBL-producing isolates carried blaTEM, 21 blaSHV, and 34 blaOXA-1-like genes. Finally, blaDHA was the most frequently observed plasmid-mediated AmpC β-lactamase gene (found in four isolates), while one isolate carried blaLAT and one blaMOX. To identify the specific bla genes detected in the PCR assays, amplicons DNA sequence analyses were performed and the results are summarized in Table 2.
Many studies on the resistance to β-lactams conducted in South America were performed using mainly a phenotypic approach. In Ecuador the few reports published on this topic lack almost totally of a genotypic study,14–17 and, to the best of our knowledge, this is one of the first studies describing the genetic characteristics of β-lactamases in this country.8,9,17
Our data confirmed the prevalence in Loja province of E. coli and Klebsiella spp. as β-lactamase producers,6,18 and a predominance of ESBL producers, showing the highest resistance levels mainly against cefotaxime, ciprofloxacin, netilmicin, and ampicillin.
It is widely documented that in South America ESBL-producing Enterobacteriaceae have one of the highest incidence in the world and CTX-Ms are the most broadly distributed.9 In particular the type CTX-M-2 is the most frequent in the Southern Cone,19 CTX-M-2-group, CTX-M-8, and CTX-M-9 in Brazil,20 CTX-M-1-group in Colombia,21 and CTX-M-2, CTX-M-14, CTX-M-15, CTX-M-24, and CTX-M-56 in Peru and Bolivia.22
In our study, it is documented that CTX-M enzymes are the dominant ESBLs in the Loja province, and among them the prevailing types are CTX-M-15 and CTX-M-14. These data are consistent with the literature21,22 as these two variants are present and quite frequent in Ecuador neighboring countries. Interestingly, the types CTX-M-55 and CTX-M-65 were detected. Actually, these variants are common in East Asia,23,24 and one possible hypothesis of their presence in Southern Ecuador can be related to the recent immigration from China and the trade relationship between Ecuador and China.25 As expected, a strong presence of TEM-type β-lactamases was also found, in particular TEM-1 β-lactamase. TEM-type presents a wide geographical distribution, and in Ecuador these data are not particularly surprising considering the incidence of TEM in neighboring Colombia26 and the trade and migratory flows between these two countries, as well as the recent intense Ecuadorian immigration to and from Europe and North America where TEM is predominant such as CTX-M-15. It is worth to mention the finding of one isolate harboring TEM-12. This ESBL variant has been already found in South America27 and it is one of the most common in North America.28 In our study, the presence of the non-ESBL OXA-1-type, SHV-1 and SHV-11, mainly found in Klebsiella (9 isolates) and Proteus (4 isolates), and the ESBL SHV-2 and SHV-12, found in Enterobacter, Klebsiella and E. coli, were also detected. The non-ESBL OXA-1-type and SHV-1 are among the most widespread in Enterobacteriaceae,29 while to the best of our knowledge SHV-11 variant is emergent in South America. Regarding ESBLs, the variants found in this study are among the most common in Latin America, indeed SHV-2 with SHV-5 were the first ESBL SHV-types described in Enterobacteriaceae,30 while SHV-12 is highly prevalent in Mexico, Colombia, and Brazil.31 Concerning AmpC β-lactamase, there is not sufficient data to draw significant conclusions on its epidemiology, but it is interesting to note the presence of two CMY-75 genes found in C. freundii. This variant has been already detected in C. freundii in Spain (EMBL/GenBank/DDBJ databases: AFK73434.1), but is emergent in South America. On the other hand DHA-1 has been previously recognized in South America, being quite predominant in Argentina.32
In conclusion, in this work the predominance of ESBLs in Southern Ecuador was demonstrated with a prevalence of CTX-M-type enzymes and we identified two emergent variants in South America. A high presence of TEM-type β-lactamases were also detected, being TEM-1 β-lactamase the most frequent variant found. However, there are some limitations in this study, including the low number of isolates and the restricted geographical area of sampling. In order to minimize these problems and gather more indicative information on molecular epidemiology and genetic characteristics of β-lactamases within the entire territory of Ecuador, further isolates from different health institutions and regions of Ecuador have to be collected and characterized.
Conflicts of interestThe authors declare no conflicts of interest.
This work was supported by the Project PROY_CCSAL_1042 of UTPL and by the Prometeo Project of the Secretariat for Higher Education, Science Technology and Innovation of the Republic of Ecuador.