Group B Streptococcus (GBS) or Streptococcus agalactiae is the main cause of death from early-onset neonatal sepsis. Because of its fulminant clinical presentation, even with the timely use of antibiotics and clinical support in the intensive care unit, the fatality rate is high and the neonatologist can do little to prevent death. In developed countries, such as the United States, the incidence of GBS infection reduced from 1.7 cases per 1000 live births in the early 1990s to 0.34 cases per 1000 live births after the implementation of antibiotic prophylaxis during labor.1
In Brazil, there is no national recommendation for antibiotic prophylaxis during labor to prevent GBS neonatal infection. The information about GBS incidence derives from two studies published a decade ago and ranged between 0.39 and 1.0 per 1000 live births.2,3 A more recent study that analyzed data from 2007 to 2011 reported a GBS incidence of 0.51 per 1000 live births.4 We describe the cases of GBS infection among infants admitted to a public neonatal intensive care unit (NICU) in Brasilia, during 2012 and 2013. The NICU has 40 beds and it is a regional reference for very low birth weight babies (<1500g) and neonatal surgery.
We identified positive blood cultures for GBS using the microbiology laboratory database. Blood culture obtained from each patient was submitted to an automated continuous monitoring system for bacterial detection (Bactec fluorescent series system® Becton Dickinson Microbiology System). The Streptococcus genus was identified by Gram stain, colony morphology, hemolysis, and the catalase test. Then, an automated panel of Gram-positive identification (MicroScan Walk-Away® Dade Behring Inc.) was used to identify Streptococcus agalactiae species. Patients’ medical records were reviewed using a standardized form to collect data.
During 2012 and 2013, 12,585 babies were born in our hospital and eight cases of GBS infection were identified. All positive cultures were collected during the first 48h of life, representing early-onset sepsis. Therefore, the incidence of GBS infection was 0.63 per 1000 live births.
The median gestational age was 33.5 weeks (range 24–41). The median number of prenatal consultations was four (range 3–9). Not a single pregnant woman had a vaginal-rectal swab collected for screening of GBS colonization and none received antibiotic prophylaxis during labor. Apart from preterm labor, three women presented risk factors for early-onset sepsis: urinary tract infection at the moment of labor or rupture of membranes longer than 18h. Labor was vaginal in five cases (62.5%) and a cesarean section in three cases (37.5%).
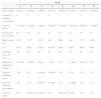
The median birth weight was 2291g (range 565–3800), the median APGAR score was seven in the first minute and eight in the fifth minute. Six newborns were male (75%) and two female (25%). No newborn had any recognizable birth defect. All presented symptoms of respiratory distress and sepsis, two just after birth and the others during the first 24h of life. All infants received ampicillin plus gentamicin. Five infants died, a case-fatality rate of 62.5%. A summary of cases is presented in Table 1
Data from 8 infants with Streptococcus agalactiae infection, Brasília, 2012–2013.
| Cases | ||||||||
|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | |
| Date of birth | 06/01/12 | 21/03/12 | 11/08/12 | 20/10/12 | 27/05/13 | 11/06/13 | 06/07/13 | 03/09/13 |
| Number of prenatal consultations | 4 | 9 | 4 | 4 | 9 | 3 | 4 | 3 |
| Type of labor | Cesarean | Vaginal | Vaginal | Vaginal | Cesarean | Cesarean | Vaginal | Vaginal |
| Gestational age (weeks) | 30 | 38 | 32 | 24 | 38 | 26 | 35 | 41 |
| Birth weight (g) | 1232 | 3475 | 1830 | 565 | 3800 | 860 | 2751 | 3200 |
| Sex | Male | Male | Female | Female | Male | Male | Male | Male |
| APGAR score 1/5min | 7/8 | 8/9 | 8/9 | 6/7 | 5/8 | 7/7 | 8/9 | 1/7 |
| Duration of membranes rupture | At labor | At labor | 6 days | 7 days | 12h | At labor | At labor | 5h |
| Time of symptom onset | 14h | 8h | At birth | At birth | 12h | 24h | 12h | 12h |
| Time of antibiotic prescription | 42h | 12h | At birth | At birth | 12h | At birth | 12h | 12h |
| Time of death | 66h | Survived | Survived | 12h | Survived | 8 days | 17h | 27h |
The GBS incidence and the high fatality rate are in line with other Brazilian studies,2–4 but higher than that achieved with antibiotic prophylaxis during labor, according to GBS prenatal screening in developed countries.1
Although the number of prenatal consultations was not low, not a single woman had been screened for GBS colonization through vaginal-rectal swab. Additionally, there were missed opportunities for antibiotic prophylaxis according to the latest Centers for Disease Control and Prevention (CDC) recommendations.1 Antibiotic prophylaxis is recommended to every preterm labor with unknown status of GBS colonization, as it was the case with four (50%) of our cases (excluding one case of a preterm infant that was born through cesarean delivery due to gestational hypertension without labor onset). However, none has received antibiotic during labor. A previous study in Brazil, had already reported 57% of inappropriate use of antibiotic prophylaxis during labor.5
In conclusion, GBS remains an important cause of early mortality among Brazilian neonates. Well-established guidelines for its prevention have been applied in developed countries for more than a decade. Nevertheless, the lack of prenatal screening and missed opportunities for antibiotic prophylaxis seem to be frequent in Brazil. Implementation of evidence-based practices is necessary in Brazil to reduce the burden of GBS and prevent death among healthy neonates.
Conflicts of interestThe authors have no conflict of interest to declare.