Treatment of tuberculosis (TB) can result in Drug-Induced Liver Injury (DILI) since hepatotoxic metabolites are formed during the biotransformation of isoniazid (INH).
DILI can be related to the genetic profile of the patient. Single nucleotide polymorphisms in the CYP2E1 gene and GSTM1 and GSTT1 deletion polymorphisms have been associated with adverse events caused by INH.
ObjectiveTo characterize the genetic polymorphisms of CYP2E1, GSTT1 and GSTM1 in TB carriers.
DesignThis is an observational prospective cohort study of 45 patients undergoing treatment of TB. PCR-RFLP and multiplex-PCR were used.
ResultsThe distribution of genotypic frequency in the promoter region (CYP2E1 gene) was: 98% wild genotype and 2% heterozygous. Intronic region: 78% wild genotype; 20% heterozygous and 2% homozygous variant. GST enzyme genes: 24% Null GSTM1 and 22% Null GSTT1. Patients with any variant allele of the CYP2E1 gene were grouped in the statistical analyses.
ConclusionPatients with the CYP2E1 variant genotype or Null GSTT1 showed higher risk of presenting DILI (p=0.09; OR: 4.57; 95% CI: 0.75–27.6). Individuals with both genotypes had no increased risk compared to individuals with one genotype.
Worldwide, tuberculosis (TB) is one biggest public health issues. In 2017, TB was estimated to have caused about 1.3 million deaths and among the top 10 causes of death in the planet, according to the World Health Organization (WHO). In Brazil, little less than 73,000 new cases were notified in 2018, representing an incidence coefficient of 34.8 cases/100,000 inhabitants. In the same year, the state of Rio de Janeiro reported an incidence coefficient of 66.3 cases/100,000 inhabitants, ranking second among states with the highest coefficient in the country. The state of Amazonas recorded the highest incidence, with 72.9 cases/100,000 inhabitants in 2018.1
In the current treatment of TB, adverse drug reactions (ADR) can vary in intensity, severity, and/or among populations.2
TB drug-induced liver injury (DILI), considered the most severe ADR, influences the current treatment with negative impact on patient treatment outcome.3
The current treatment of TB in Brazil,4 although effective, can induce adverse events, including DILI.3
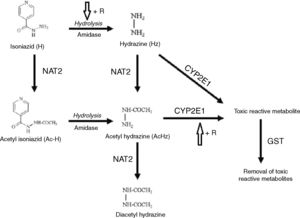
Isoniazid (INH) is the main contributing drug for DILI development. It is metabolized by CYP2E1 and generates hepatotoxic metabolites. The GST enzyme reduces this toxicity because it can catalyze the conjugation reaction of the sulfhydryl terminal of these compounds (Fig. 1).5,6
Route of biotransformation of isoniazid (INH). R: Rifampicin. Adapted from Sotsuka et al.6 The use of this figure has been authorized by the copyright owner.
CYP2E1 is expressed by CYP2E1, a polymorphic gene. In this study, four alleles of the gene were characterized. The CYP2E1*1A wild type allele presents a cleavage site for the Dra I and Rsa I restriction enzymes by polymerase chain reaction-restriction fragment length polymorphism (PCR- RFLP). The variant alleles are CYP2E1*5B, with polymorphism in the promoter region, with no site for Rsa I; CYP2E1*6, with polymorphism in intron 6, with no site for Dra I; and CYP2E1*5A, with polymorphism in the promoter and intronic regions, has no site for any of these two enzymes. It is not clear which allele has greater influence on the increased activity of CYP2E1.7
GST enzymes play a protective role on the mechanism of liver detoxification, catalyzing glutathione reaction, facilitating solubilization, and excretion of toxic substrates.8 The GSTM1 and GSTT1 genes present polymorphisms by partial or total deletion. Individuals with total deletion are more prone to toxic effects because the enzyme activity is absent; in partial deletion, it can display some activity.3
The objective of this study was to evaluate the genotype and the allele frequencies of the CYP2E1, GSTM1 and GSTT1 gene variants besides investigating the relation between these genetic polymorphisms and the development of DILI in patients treated for active pulmonary tuberculosis, admitted to the Hospital Estadual Santa Maria (HESM) [Santa Maria State Hospital], in the state of Rio de Janeiro, Brazil.
Materials and methodsType of studyThis was an observational prospective cohort study of patients treated for active pulmonary tuberculosis. The population comprised hospitalized adult patients receiving care at Santa Maria State Hospital (HESM), in the state of Rio de Janeiro, Brazil. This study was part of the project Desnutrição e Imunogenética na Tuberculose e Infecção pelo HIV [Malnutrition and Immunogenetics in Tuberculosis and HIV Infection], approved by the Ethics Committee of the University Hospital Clementino Fraga Filho under the number 004/05.
Brazilian Guidelines recommends the current treatment of TB to be conducted in outpatient clinics and directly observed. Hospitalization is recommended in some cases, such as social vulnerability and absence of a permanent residence.9 Patients in this study were followed up during their hospitalization. TB drugs were taken daily, at the same time, and observed by a health professional.
Inclusion criteria: age over 18 years; diagnosis of active pulmonary TB according to the Programa Nacional de Controle contra TB (PNCT) [National Control Program against TB] criteria;9 starting treatment according to the current treatment adopted by the PNCT9 together or in a period close to hospitalization; and agreeing to participate in the study through understanding and signing of the Informed Consent form. Exclusion criteria: pregnancy; neuromuscular disorders; chronic liver disease or liver failure; presence of intolerance/resistance to drugs of the current regimen adopted by PNCT9 in the period of data collection of the biological material for the study; use of other potentially hepatotoxic drugs; and positive serology for acute infection by hepatitis A, B and/or C.
Sample collectionFrom August/2012 to September/2013, 49 patients were included in the study. Before starting treatment, blood tests were performed (alanine transaminase, aspartate transaminase, alkaline phosphatase, gamma-glutamyl transferase, total and fractions bilirubin, total and fractions protein, complete blood count and markers for viral hepatitis A, B and C). DNA was extracted from total blood. During hospitalization, venous blood was collected to evaluate patients’ liver function every 10 days beginning at admission (considered day zero) until close to the 40th day.
DNA extraction and genotypingGenomic DNA was extracted according to the manufacturer’s guidelines (Promega - The Wizard Genomic DNA Purification Kit). PCR - RFLP was performed to detect polymorphisms in the CYP2E1 gene in the promoter and intronic regions (with the Rsa I and Dra I restriction enzymes, respectively), with protocol adapted from YANG et al.10 The GSTM1 and GSTT1 genotyping was performed using Multiplex PCR with protocol adapted from HONMA et al.11 To validate the genotyping techniques, five samples were sequenced (in BigDye Terminator v3.1 Cycle Sequencing Kit) and 5/5 samples genotypes were confirmed.
Adverse reactions to drugsDILI due to TB treatment was defined by the presence of: ALT two-fold higher than the upper limit of normal (ULN) and/or AST two-fold higher than the ULN, together with increased total serum bilirubin greater than 1.3mg/dL.12
The genetic profile of the patients was identified blind to DILI identification. For ADRs evaluation, clinical and nutritional data collection and clinical records were used. For DILI evaluation, only the ADRs related to treatment of TB were considered;13 each ADR was counted only once per patient and no ADR was classified according to intensity.
Statistical analysisData were entered in the Excel 2007 spreadsheet and descriptive statistical analysis of the study sample characteristics was conducted with the tools of the same software.
Hardy–Weinberg’s equilibrium was calculated to evaluate the correlation of the frequencies found for CYP2E1 genotypes.14
Groups with and without DILI were comparison using non-parametric Wilcoxon’s test, Pearson’s chi-square and Fisher’s exact test.
For the final analysis, logistic regression was conducted to estimate the crude and adjusted odds ratio with a 95% confidence interval, using the statistical software R (Version 3.2.2).
Patients with any CYP2E1 gene variant allele were grouped for the purpose of statistical analyses. As the sample size was small (n=45), variables reaching p<0.10 were considered statistically significant.
ResultsStudy sampleTable 1 shows the characteristics of the pulmonary TB patients included in the study, grouped in patients with DILI (DILI Group) and without DILI (No-DILI Group). Of the 49 patients, four were excluded: two were drug resistant/ intolerant to the current TB treatment; one had a unspecific pneumonia, and one had acute phase antigens for hepatitis B. A total of 45 patients completed the study and had DNA extracted and genotyped for CYP2E1, GSTT1 and GSTM1.
Demographic and clinical characteristics of patients under treatment of TB included in the study.
| Characteristics of patients | ||||
|---|---|---|---|---|
| All Patients (N=45) | Patients without DILI (N=39) | Patients with DILI (N=6) | ||
| Gender (%) | ||||
| Male | 67 | 72 | 33 | |
| Female | 33 | 28 | 67 | |
| Age – years, mean (+ or − SD) | 42.0 + (15.5) | 42.0 + (15.5) | 46.8 + (16.3) | |
| Male | 44.5 + (15.2) | 44.1 + (15.5) | 46.5 + (12.0) | |
| Female | 37.1 + (15.4) | 33.5 + (12.6) | 47.0 + (19.8) | |
| Tobacco (%) | 82 | 90 | 33 | |
| Alcohol (%) | 76 | 85 | 17 | |
| Illicit drugs (%) | 58 | 62 | 33 | |
| HIV (%) | 11 | 8 | 33 | |
| Hospitalization time – days, mean (+ or − SD) | 107.6 + (69.4) | 105.6 + (58.7) | 120.7 + (67.0) | |
| Hospital discarge (% cure/improved) | 75 | 72 | 100 | |
| RAM/paciente | 3 | 2 | 5 | |
%: percentage of total of patients; SD: Standard Deviation; g/dL: grams per liter; ADR: Adverse Drug Reaction.
The majority of the patients (67%) were males, mean age 42 years (18–82 years). A total of 82% were active smokers and 76% consumed alcohol. All alcohol consumers were smokers; 58% used illicit drugs and 11% were infected by HIV. The average hospitalization time was 110 days; the reasons for hospital discharge were death - three patients (7%); cured – nine (20%); improved - 25 (56%), and discharge for indiscipline - eight (18%). During the hospitalization period, patients had mild anemia. A total of 16% of the studied population showed one type of ADR, 60% showed two or more types, with an overall mean of three ADR/patient.
Genotypic distributionTable 2 shows the genotypic sample distribution in the Promoter Region of the CYP2E1 gene (C1053T; rs2031920): 98% of patients had wild genotype (CYP2E1*1A/1A or c1c1) and 2% heterozygous genotype (CYP2E1*1A/5B or c1c2); no homozygous variant genotype for this region (CYP2E1*5B/5B or c2c2) was found. In the Intronic Region (T7632A; rs2070673), 78% were wild genotype (CYP2E1*1A/*1A or DD), 20% heterozygous genotype (CYP2E1*1A/*6 or DC), and 2% homozygous variant genotype (CYP2E1*6/*6 or CC). Null GSTT1 genotype was found in 22% of the patients and Null GSTM1 in 24%.
Genotypic and allelic distribution of CYP2E1, GSTM1 and GSTT1 of patients under treatment of TB included in the study.
| Frequency | CYP2E1 | |||
|---|---|---|---|---|
| Promoter Region | Intronic Region | |||
| Genotypic | c1c1 | 0.978 | DD | 0.778 |
| c1c2 | 0.022 | DC | 0.200 | |
| c2c2 | 0 | CC | 0.022 | |
| Allelic | c1 | 0.989 | D | 0.878 |
| c2 | 0.011 | C | 0.122 | |
| GST | ||
|---|---|---|
| Null GSTT1 | Null GSTM1 | |
| Frequency | 0.22 | 0.24 |
c1: Wild allele of the promoter region; c2: Variant allele of the promoter region; D: Wild allele of the intronic region; C: Variant allele of the intronic region.
A total of 13% (6/45) of the patients were diagnosed with DILI according to study criteria. Of these, 33% were smokers, 17% consumed alcohol and 33% were using an illicit drug; 33% were HIV infected; 100% of this group were discharged from hospital as cured or clinically improved.
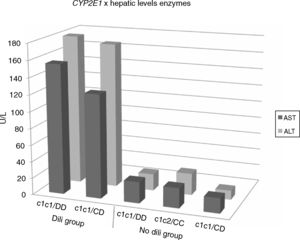
A mean of two ADR/patient was observed in the No-DILI Group and five in the DILI Group, in addition to the elevation of the liver enzymes levels. The DILI Group proved to be more sensitive to anti-TB drugs. The five TB/HIV co-infected patients showed greater sensitivity to the drugs like the DILI Group, with a mean of five ADR/patient. Table 3 shows the characteristics of both groups. For the CYP2E1 gene, 50% of the DILI Group had the wild genotype (c1c1/DD) and 50% had the heterozygous genotype. In the No-DILI Group, higher frequency of the wild genotype was observed. The DILI Group had a higher frequency of the Null GSTT1 genotype, while the No-DILI Group had a higher frequency of the Null GSTM1.
Distribution of the genotypic, demographic, clinical and laboratory data of the DILI and No-DILI Groups in the studied sample.
| Anti-Tuberculosis Drug-Induced Liver Injury | |||
|---|---|---|---|
| Presence | Absense | p-value | |
| CYP2E1 | |||
| c1c1/DD | 3/6 | 32/39 | |
| c1c2/CC orc1c1/DC | 3/6 | 7/39 | 0.09*‡ |
| GST | |||
| NULL T1 | 3/6 | 7/39 | 0.09* |
| NULL M1 | 1/6 | 10/39 | 0.6 |
| ALT (U/L) | 176.3 | 20.7 | <0.001* |
| AST (U/L) | 139.7 | 25.1 | <0.001* |
| T BIL (mg/dL) | 1.18 | 0.58 | <0.003* |
| Age (years) | 46 | 41 | 0.39 |
| Sex | 0.06* | ||
| Male | 2 | 28 | |
| Female | 4 | 11 | |
| Hospitalization Time (days) | 120.7 | 105.6 | 0.59 |
c1 and D: Wild alleles; c2: Variant allele of the Promoter Region; C: Variant allele of the Intronic Region; ALT: Alanine aminotransferase; AST Aspartate aminotransferase; U/L: Unit per liter; T BIL: Total bilirubin; ULN: Upper limit of normal; F: Female sex; M: Male sex; * statistically significant; ‡ Fisher’s exact test.
The DILI Group was discreetly older and had longer hospitalization time than the No-DILI Group, with no statistical significance. The former had more females than the latter (p<0.06).
The DILI Group showed serum level four-fold higher (p<0.001) three-fold higher (p<0.001) than the ULN for ALT and AST, respectively. The No-DILI Group maintained normal enzymatic levels. Fig. 2 illustrates the serum levels of liver enzymes related to their genotypes in the studied sample.
Logistic regression analysisTable 4 shows the results of the logistic regression analysis. Patients with at least one CYP2E1 variant genotype or with Null GSTT1 genotype had an odds ratio 4.57 times higher than the other patients (p=0.09). Other factors that could be associated with the risk of developing DILI, such as the use of illicit drugs and/or the presence of the GSTM1 genotype, were not statistically significant.
Association of the studied genotypes and variables of the anti-tuberculosis drug- induced liver injury treatment in the samples of the patients under treatment of TB included in the study.
| Variable | OR (95% CI) | p-value (<0.10) |
|---|---|---|
| CYP2E1 Variant | 4.57 (0.75–27.6) | 0.09* |
| NULL GSTM1 | 0.58 (0.06–5.58) | 0.6 |
| NULL GSTT1 | 4.57 (0.76–27.6) | 0.09* |
| Use of illicit drugs | 0.39 (0.06–2.36) | 0.3 |
Every patient who had at least one variant allele, regardless of the region, due to low frequency, was considered a variant genotype of CYP2E1. A p-value < 0.10 was considered significant due to the reduced number of patients included in the study.
In this study, 13% of patients presented DILI, similar to studies that included viral hepatitis in their exclusion criteria (EC). Compared to other studies, the frequency of DILI can vary. In the Brazilian population, DILI frequency seems to be related to the rigor of the EC used by the authors. Apparently, these predisposed conditions can cause a selection bias in the study and be a contributing factor for higher rates of DILI. Studies that were less rigorous and included active smokers, patients with abusive alcohol consumption, and patients co-infected with HIV and viral hepatitis (B and/or C) reported higher DILI frequency. Therefore, including patients who have predictive factors for DILI during treatment of TB will likely have higher rates of DILI, as reported by Zaverucha et al.15 who found 40% and Monteiro et al.16 who found 33%. Other Brazilian studies (Vieira & Gomes;13 Possuelo et al.;17 Teixeira et al.;18 and Santos et al.19) characterized by more restrictive eligibility criteria (with no predictive factors for DILI), have found lower DILI frequencies (<20%). Liu et al.20 have argued that these factors influence the risk and severity of DILI.
The American Thoracic Society21 (ATS) criteria for defining DILI are more rigorous than Bénichou criteria.12 Among the parameters used, liver enzymes level should be five-fold higher than the ULN, while Bénichou12 parameters set up a level of two-fold. One would expected to find a relationship between the different criteria used to define DILI and its frequency in the analyzed studies. However, the choice of DILI definition had no impact on the frequency of DILI.
In Brazilian studies (including the current study) the variation found in the DILI frequency as per GST genes was small. Monteiro et al.16 have suggested that the Null GSTM1 gene is probably associated with the intensity of liver toxicity. Teixeira et al.18 have found no association between GST polymorphisms and risk of DILI. Singla et al.5 have argued that patients with deletion in both GST genotypes have greater risk of DILI. In contrast, Tang et al.3 have not associated the deleted GST genotypes with the risk of DILI.
Some authors have reported that patients with DILI have higher frequency of the CYP2E1 variant alleles. In our study, patients with the presence of CYP2E1 variant alleles are at higher risk for DILI. In the literature, the findings of the Brazilian population show some discrepancies regarding the association of CYP2E1 genotype and risk for DILI. Zaverucha et al.15 found an increased risk for DILI in individuals with CYP2E1 variant alleles. However, Teixeira et al.18 did not find any relationship between DILI and the CYP2E1 genotype.
Deka et al.22 have found a significant association of the two variant alleles (c2 and C) with DILI. Bose et al.23 have found a significant association of the C variant allele with DILI in patients with viral hepatitis. A significant association of the c2 allele with DILI was reported by Singla et al.5 Thus, it has been suggested that CYP2E1 variant genotypes could be associated with increased risk of DILI during anti-TB treatment.
Roy P, Majumder & Roy B2 and Ramachandran & Swaminathan7 have proposed that INH can induce CYP2E1 activity in mice, an activity dependent on the plasma concentration of INH. On the other hand, INH can inhibit CYP2E1 activity, a fact more common in individuals of variant genotype. In addition, they have related the increase in enzyme activity to higher production of hepatotoxins, which increases the risk for DILI. Ramachandran & Swaminathan7 have pointed to the important existing discussion in the literature about which allele has greater influence on the increased activity of CYP2E1, variant or wild.
Most of the research subjects were male. According to data from the Sistema Único de Saúde (SUS) [Unified Health System]24 during the inclusion period, 230 patients were admitted to the HESM with active pulmonary TB, 72% males and 28% females. Data are consistent with the Brazilian TB cases according to the PNCT25 Epidemiological Bulletin that describes a two-fold higher incidence coefficient in males. The studied sample consisted of patients in productive age, mean of 44 years for males and 37 years for females.
Patients who had cure discharged remained in the hospital until completing treatment (about 180 days), while the other patients remained hospitalized for a shorter period. There was no follow-up of patients’ treatment after hospital discharge, a fact that could be considered a study limitation. The mean time of patients’ hospitalization was 61% shorter than the time needed to complete treatment. Mild anemia found in patients during hospitalization is compatible with anemia of chronic diseases, common in Tuberculosis.26,27
Among the 45 participants, five (11%) were co-infected with HIV, similar to that described by the PNCT28 Epidemiological Bulletin that reports a 10% rate of HIV co-infection among TB cases of Rio de Janeiro (RJ) state.
The ADR frequency found in this study is slightly higher (3 ADR/patient) than that reported by Vieira & Gomes13 who have pointed out that 49% of their patients had one or more ADR associated with TB treatment. The discrepancy can be explained by the characteristics of the studied population: no permanent residence, poor nutritional status and compromised immune system, besides having limited access to basic sanitation and hygienic conditions. The DILI group showed predominance of the female sex and an increased sensitivity to anti-TB drugs when compared to the No-DILI group. The studied sample is consistent with findings in the literature: higher DILI frequency among females, malnourished individuals with pre-existing liver disease and advanced age.29 The studied sample showed some of these characteristics except for prior liver disease, since it was one of the exclusion criteria. Piva et al.29 have observed that the rate of treatment default among malnourished patients was three-fold higher than what is acceptable because malnourished patients tend to have a compromised immune response more often than the normal weight patient.
This study had a small sample size (n). However, the other works cited had periods of inclusion greater than 13 months. Monteiro et al.16 have included 177 patients over five years, Zaverucha et al.15 have included 131 patients over seven years and Teixeira et al.18 have included 167 patients over 10 years. Even with reduced sample size, we have observed an important effect in the evaluation of the risk for DILI.
ConclusionPatients who have the CYP2E1 variant genotype or Null GSTT1 have a four-fold greater risk of having DILI during anti-TB treatment. Those who have both the CYP2E1 variant genotype and Null GSTT1, have no increased risk of having liver injury compared to the individuals who have only one of these genotypes. More studies with larger sample sizes should be conducted for a better understanding of the role of these genetic factors in the association with the occurrence of DILI during treatment of TB.
The authors thank the colleagues who contributed to the enrichment of this study: the teams of the Laboratory for Clinical Analyses of the Faculty of Pharmacy, of the Laboratory of Microbiological Quality Control of Drugs/Medicines, Food and Cosmetics and of the Laboratory of Molecular Virology, all of the Universidade Federal do Rio de Janeiro. Acknowledgments are extended to the financial support of Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ) [Foundation for Research Support of the State of Rio de Janeiro], without which the development of this work would not have been possible.