COVID-19 pandemic has been a problem worldwide. It is important to identify people at risk of progressing to severe complications and to investigate if some existing antivirals could have any action against SARS-CoV-2. In this context, HIV-infected individuals and antiretroviral drugs might be included, respectively. Herein we present the case of a 63-year-old HIV-infected woman with undetectable viral load, on dolutegravir, tenofovir and lamivudine, who was hospitalized due to COVID-19 pneumonia. In spite of having some clinical markers of severity on admission, the patient improved and was discharged after a week. To our knowledge, this is the first report of severe SARS-CoV-2 infection in an HIV-infected individual in Brazil.
COVID-19, caused by the severe acute respiratory syndrome coronavirus type-2 (SARS-CoV-2), has been declared a pandemic in March 2020, following the first case reported in Wuhan, China in December 2019. Some patients, such as those suffering from cardiovascular disease, diabetes mellitus and obesity, were soon identified at greater risk for worse clinical outcomes. Nevertheless, there is still a lot to be discovered regarding this disease. In this context, not only the risk of COVID-19 complications among people living with HIV (PLWH) remains uncertain, but also the potential protective benefits of antiretroviral (ARV) drugs.
Despite of more than 12 million cases of COVID-19 worldwide as early July, reports of HIV/Sars-CoV-2 co-infection are still uncommon,1 with the first known case in the USA only being reported on May 22, 2020,2 People living with HIV accounted for only 1% of 16,749 patients with COVID-19 hospitalized in the United Kingdom, on a large prospective observational cohort study, with HIV having no impact on survival.3 Thereafter, the prognosis of PLWH after the diagnosis of COVID-19 has also been a subject of debate.4 While some researchers believe that HIV immunosuppression could result in greater susceptibility to SARS-CoV-2 infection,5 others believe that these patients would be at a lower risk of complications, since impairment in cellular immunity might be associated with less inflammation,6 reducing the incidence of cytokine storm which has been associated with more severe cases of COVID-19.
There is also some controversy about the potential advantages against SARS-CoV-2 from being on ARVs.7 There is some in vitro evidence that these drugs could have an impact against this virus.8,9 Herein we present the case of an HIV-infected woman on antiretroviral therapy who was hospitalized at our center with COVID-19 pneumonia.
Case reportA 63-year-old woman was admitted to COVID-19 Unit at Hospital de Clinicas de Porto Alegre (HCPA), on southern Brazil, on May, 2020. The patient had been diagnosed with HIV infection in 2005, and was on tenofovir (TDF), lamivudine (3TC), and dolutegravir (DTG) since November 2019 (before she was on atazanavir/ritonavir, switched for DTG). Her viral load had been undetectable for a long time and her CD4+ cell count was 426 cells/mm3 (CD4/CD8 ratio 1.25). Systemic arterial hypertension (SAH), well controlled with hydrochlorothiazide and losartan, was the other comorbidity she had.
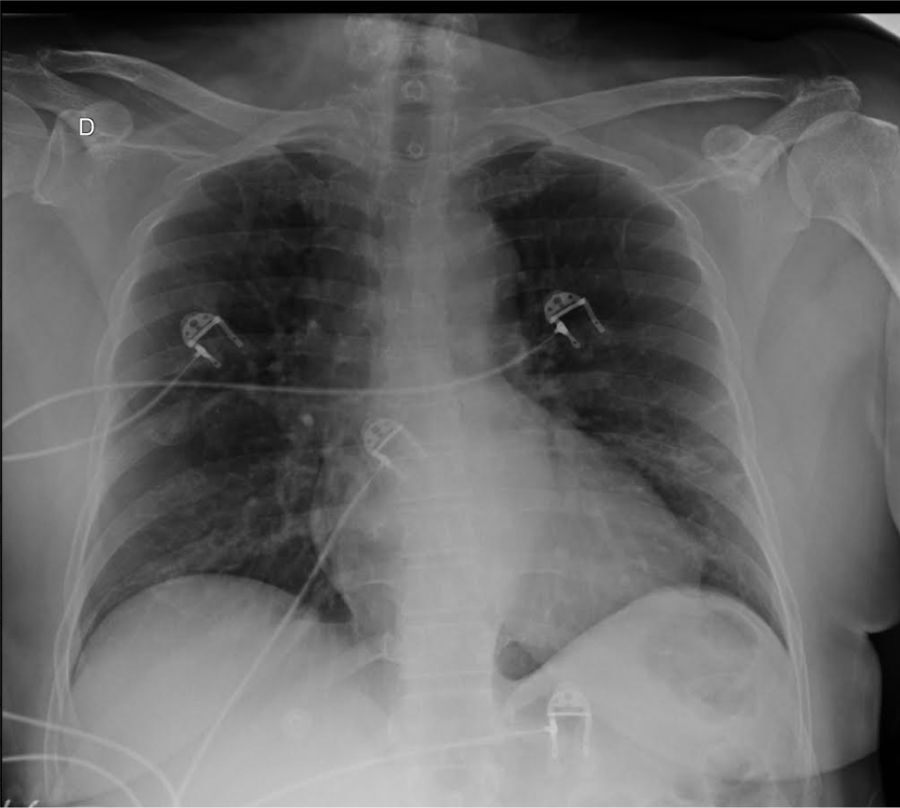
At presentation, she complained of fever (39 °C), myalgia, inappetence, nausea, abdominal pain, diarrhea, hyposmia and hypogeusia, in addition to cough and dyspnea for a week. Upon admission, white blood cells (WBC) count was 9250 cells/mm3 (76% neutrophils, and 16% lymphocytes). She had elevated C-reactive protein (65.5 mg/L), total creatine kinase (307 U/L) and lactate dehydrogenase (316 U/L). Serum creatinine was 0.84 mg/dL and she had no abnormalities in clotting tests, including a D-Dimer level of 0.42 ug/mL (with our reference value being up to 0.5 ug/mL). First arterial blood gas analysis (ABG) was performed with the patient on supplemental oxygen through a nasal cannula, but she did not present hypoxemia, with a 107 mmHg pO2 and a peripheral oxygen saturation (SpO2) of 98.5%. Chest X-ray exhibit opacities in middle thirds of both lungs (Fig. 1). Sars-CoV-2 was detected by RT-PCR in nasopharyngeal secretion swab.
The patient was treated with supportive measures that included oxygen via nasal cannula and received antibiotic therapy with amoxicillin/clavulanate for a total of seven days. After giving consent, she was randomized to one of the arms of a randomized clinical trial (Coalition-1 trial10) and received hydroxychloroquine 400 mg bid plus azithromycin 500 mg per oral according to trial protocol, with no side effects related to any of these therapies during this period. Even though she presented some severity markers on admission, she had a favorable clinical evolution, with no need for treatment in ICU or requiring more invasive forms of oxygen therapy, being discharged in good clinical conditions seven days after hospitalization.
ConclusionTo our knowledge, this is the first reported case of COVID-19 disease in an HIV-infected individual in Brazil and remains the only one that we had the opportunity to care. This finding is of special interest, as Porto Alegre, the capital of the southernmost state of Brazil (Rio Grande do Sul), is one of the cities with highest HIV incidence in Brazil.11 This report has the purpose to illustrate the several peculiarities of SARS-CoV-2 infection worldwide. Although the patient presented a severe course of COVID-19, the ARVs she was taking did not protect her from acquiring the infection.
Among the drugs used to treat HIV, lopinavir/ritonavir was the first to assessed against SARS-CoV-2. In spite of the studies in 2003 during SARS epidemic showing a decrease in mortality, intubation rates and unfavorable outcomes on SARS with the use of lopinavir/ritonavir,12 a recent clinical trial showed no benefit of this medication for the treatment of severe cases of COVID-19.13 Other combinations of protease inhibitors are under testing, with atazanavir/ritonavir showing greatest inhibitory potential in vitro against SARS-CoV-2.8 A clinical trial of darunavir/cobicistat for treatment of COVID-19 is currently on progress in China.14 Other ARVs have also shown some activity in vitro and in animal models. DTG, an integrase inhibitor, had demonstrated activity against SARS-CoV-2.8,9,15,16 Likewise, TDF, a nucleotide analogue reverse transcriptase inhibitor widely used in the treatment of HIV and Hepatitis B infections, has emerged as a new investigative agent against COVID-19. It is from the same class as remdesivir, a novel nucleotide analogue that has activity against SARS-CoV-2 in vitro17 and has been recommended for hospitalized patients with severe COVID-19 in many guidelines.18 However, no ARV has demonstrated clinical impact so far.
One of the first published case series, comprising of 33 patients hospitalized for COVID-19 followed up at HIV centers in Germany, suggested that although SARS-CoV-2 infection could occur during treatment with ARVs, these patients did not appear to be at risk for worse clinical outcomes among those with symptomatic COVID-19.19 A recent large Spanish cohort study of 77,590 HIV-infected people on ARVs identified similar risk factors for hospitalization, admission to ICU, and death when compared to the general population.20 However, HIV-infected patients on TDF/FTC had a lower risk of COVID-19 and related hospitalization than those receiving other therapies. Whether the difference observed is due to the profile features of patients using this ART or the direct antiviral effect of the drug is still a matter of debate.20 Currently, one trial combining tenofovir-alafenamide/emtricitabine and lopinavir/ritonavir to treat COVID-19 patients and another with tenofovir/emtricitabine as pre-exposure prophylaxis against COVID-19 in health care workers are on progress.21 In our patient, these ARVs did not prevent SARS-CoV-2 infection. Still, a lot is still to come regarding this coronavirus infection and the clinical course in HIV-infected individuals and the possible impact of ARVs.