To study whether patients with HIV-1 associated lipodystrophy (LD) on highly active antiretroviral treatment (HAART) have more psychopathology and worse psychosocial adjustment than a similar group without this syndrome.
MethodsIn a cross-sectional, observational study we compared 47 HIV-1 infected patients with LD (LD group) with 39 HIV-1 infected patients without LD (non-LD group). All participants were on HAART. The Beck Depression Inventory (BDI), the State and Trait Anxiety Inventory (STAI) and the Goldberg Health Questionnaire (GHQ-60) were administered. Levels of familial, work and social adjustment and adjustment to stressful events were evaluated in a semi-structured interview. Clinical information was extracted from the clinical records.
ResultsIn the univariate analysis patients with LD showed higher state anxiety scores (p=0.009) and worse work adjustment (p=0.019) than those without LD. A total of 45.3% of LD patients scored above the cut-off point on the trait anxiety scale, and over 33.3% scored above the cut-off point on the BDI, GHQ and state anxiety scales. However, in multivariate analyses LD was not independently associated with psychopathology or with worse adjustment in the studied areas.
ConclusionsThe finding that LD was not a predictor of greater psychopathology or worse psychosocial adjustment in HIV-1 infected patients, despite the high scores found, suggests that factors not taken into account in this study, such as LD severity and self-perception should have been included in the analysis. Further studies including a greater number of variables and a larger sample size will advance our understanding of this complex condition.
HIV mortality and morbidity have decreased dramatically since the availability of HAART,1 but as life expectancy increases new metabolic complications have emerged. One of these complications is lipodystrophy (LD), also referred to as the HIV-lipodystrophy syndrome or fat redistribution syndrome. It was first described in 19982 but no standard definition has yet been described. LD involves elements related to metabolic disorders and redistribution of body fat. The most important clinical feature is the fat loss (lipoatrophy) from the face, limbs and buttocks, sometimes with central fat accumulation on the abdomen, breasts and dorso-cervical spine. These body changes, especially those in the face, are the most frequent and distressing signs of this clinical syndrome.
Patients perceive body changes, especially facial lipoatrophy, as a stressful and disfiguring symptom of their disease.3,4 They fear that fat loss represents progression of the disease and worry that visible changes may lead to unintentional disclosure of their HIV status.5 Consequently, physical manifestations of LD affect self-esteem, self-image and self-perception, and increase feelings of stigmatization.3,6,7 This in turn may cause psychological distress and negatively affect social, professional and sexual relationships,3,5,7 leading to limited participation in physical activities, poor social functioning, loneliness and isolation.4,7,8
Psychopathology and psychosocial adjustment has been studied previously but findings are controversial. Collins et al.3 and Power et al.,7 for example, investigated quality of life and psychosocial adjustment in HIV-1 positive patients with LD and found that anxiety and depression were higher than in non-LD patients. On the other hand, Preau et al.9 found LD to be independently associated with depression but not with anxiety. However, Steel et al.10 studied the effects of LD on quality of life and depression in HIV-infected men on HAART but did not find any association between LD and depression. Much of this controversy may be due to differences in population samples and methodologies: study samples were mainly small and non-homogenous, and the methods used varied from qualitative to quantitative approaches.
Psychopathology and psychosocial adjustment in HIV infected patients with LD compared to those without LD therefore remains unclear. Furthermore, no studies have yet been performed using validated tools in the Spanish population.
The purpose of this study was to determine whether psychopathology and psychosocial adjustment in HIV-1 infected patients on HAART who developed LD differed from HIV-1 infected patients without this syndrome.
Materials and methodsWe conducted a cross-sectional study of patients receiving care at our department. The study was carried out in the Infectious Diseases Unit and the Psychosomatic Unit at the Hospital de la Santa Creu i Sant Pau, Barcelona.
All HIV positive patients on HAART receiving care at the outpatient service were invited to participate in the study. The objectives of the study were explained and patients who signed the informed consent were referred to the study psychologist. During the interview with the psychologist all inclusion and exclusion criteria were reviewed. The study was approved by the institutional ethics committee. Participants’ anonymity and confidentiality were ensured at all times.
The study population included literate female and male patients, 18 years and older, who were HIV-1 infected and on HAART. We excluded patients without the necessary Spanish language skills to answer the questionnaires and those who had any physical, psychiatric or cognitive disorders that could interfere with their participation in the study, or compromise the reliability of the self-reported measures. We also excluded patients with missing data.
Ninety per cent of patients agreed to participate in the study and signed the informed consent form. The sample consisted of 86 patients: 47 clinically evident LD and 39 non-LD patients. As there is no objective, validated, sensitive and specific case definition of HIV-associated LD,2 the LD syndrome was considered present when both physician and patient agreed that signs and symptoms of the disorder were present.11
The psychologist conducted a semi-structured interview where data related to sociodemographic factors were obtained. Psychological tests (GHQ, BDI, STAI) were self-administered and clinical variables were abstracted from medical records.
Demographic characteristicsDemographic variables collected were age, gender, cohabitation status, number of years living outside country of birth if immigrant, educational level, profession and employment status. Participants were asked about familial, work and social adjustment, adjustment to stressful events, and perceived familial and social support in order to establish their psychosocial adjustment. Questions were as follows: “Are you satisfied with your adjustment to new life situations?” “Are you satisfied with your social adjustment?” “Are you satisfied with the support of your friends?” All questions were open-ended: “yes” (indicating adjustment) or “no” (indicating maladjustment).
The General Health Questionnaire (GHQ)The General Heath Questionnaire12 evaluates general mental health. It has been used by other authors in studies in HIV infected patients.13,14 In this study we used the Spanish validated version of this questionnaire.15 It was self-administered and scores above 11 were considered pathological.
The Beck Depression Inventory (BDI)The Beck Depression Inventory16 assesses depression on a 21-item scale. In positive cases, it also evaluates severity and intensity of the symptomatology. It has been successfully used in HIV infected patients in previous studies.17–20 In this study the Spanish validated version was used.21 The questionnaire was self-administered and scores of 10 or above were considered indicative of depression.
The State and Trait Anxiety Inventory (STAI)Anxiety was assessed by using the State and Trait Anxiety Inventory.22 This tool is composed of two scales, “State anxiety” that describes feelings indicative of current anxiety, and “Trait Anxiety” which describes feelings indicating normal anxiety. Both scales have 20 items and the answers to all items are scored from 0 (no anxiety) to 3 points (highest anxiety). Scores were transformed into percentiles and those above the 55th percentile were considered pathological. We used the Spanish validation carried out by Bermúdez23 Several authors have used this inventory in HIV studies.24,25
Clinical variablesData related to HIV infection and clinical status were also recorded. These variables included years since HIV-1 diagnosis, years on HAART, CD4 counts (cels/mm3), plasmatic HIV viral load (logcopies/ml), and mode of HIV acquisition.
Statistical analysesMeans and standard deviation (SD) were obtained for quantitative variables and percentage and frequencies for categorical data. The Student t-test was used to compare quantitative variables between groups (with and without LD). The corrected results were used to avoid possible heteroscedasticity. Fisher's exact test was used to compare categorical values. A logistic regression model using a forward procedure was used to predict the presence of psychopathology. The dependent variables were psychopathology (measured with the GHQ), trait anxiety and state anxiety (estimated with the STAI), and depression (evaluated with the BDI). The independent variables were LD, educational level, employment status, familial adjustment, social adjustment, work adjustment, adjustment to stressful events, profession and the questions asked in the interview.
A bilateral approach was used for all calculations and the probability of a type I error was 5% (α=0.05). Values of p<0.05 were considered significant. All the analyses were done using the SPSS statistical package v 14.0.
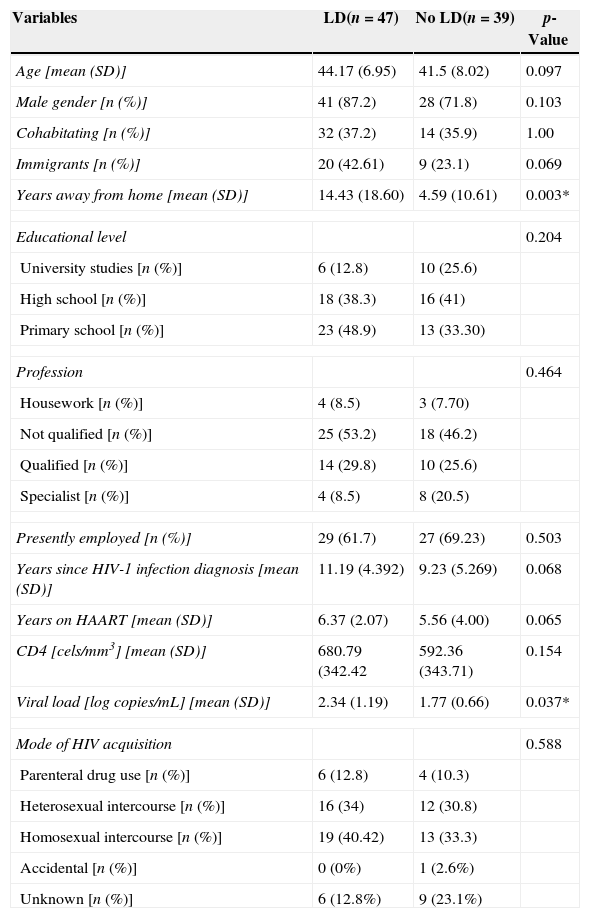
ResultsNinety-six patients were approached and 90% (n=86) agreed to participate in the study. All participants gave written informed consent. Mean age was 42.9 (range: 24–62) years and 80.2% were men. Those with LD were on average three years older than non-LD patients (p=0.097) but differences were not significant. A total of 39.5% of the participants had completed high school, 65.1% were presently employed and 27.9% were qualified workers. More than 33% were immigrants. The percentage of immigrants was almost 20% higher in the LD group (p=0.069) but this was not statistically significant.
HIV infection was mainly transmitted by sexual intercourse and no “mode of acquisition” differences were found between groups. Patients with LD showed statistically significant higher values of viral load (p=0.037). No statistical differences were found between LD patients who were diagnosed some years earlier (p=0.068) and were on HAART regimen for a longer period (p=0.065) and non-LD patients. Sociodemographic and clinical variables are shown in Table 1.
Sociodemographic and clinic characteristics of LD and non-LD groups [n=86; *p<0.05].
| Variables | LD(n=47) | No LD(n=39) | p-Value |
|---|---|---|---|
| Age [mean (SD)] | 44.17 (6.95) | 41.5 (8.02) | 0.097 |
| Male gender [n (%)] | 41 (87.2) | 28 (71.8) | 0.103 |
| Cohabitating [n (%)] | 32 (37.2) | 14 (35.9) | 1.00 |
| Immigrants [n (%)] | 20 (42.61) | 9 (23.1) | 0.069 |
| Years away from home [mean (SD)] | 14.43 (18.60) | 4.59 (10.61) | 0.003* |
| Educational level | 0.204 | ||
| University studies [n (%)] | 6 (12.8) | 10 (25.6) | |
| High school [n (%)] | 18 (38.3) | 16 (41) | |
| Primary school [n (%)] | 23 (48.9) | 13 (33.30) | |
| Profession | 0.464 | ||
| Housework [n (%)] | 4 (8.5) | 3 (7.70) | |
| Not qualified [n (%)] | 25 (53.2) | 18 (46.2) | |
| Qualified [n (%)] | 14 (29.8) | 10 (25.6) | |
| Specialist [n (%)] | 4 (8.5) | 8 (20.5) | |
| Presently employed [n (%)] | 29 (61.7) | 27 (69.23) | 0.503 |
| Years since HIV-1 infection diagnosis [mean (SD)] | 11.19 (4.392) | 9.23 (5.269) | 0.068 |
| Years on HAART [mean (SD)] | 6.37 (2.07) | 5.56 (4.00) | 0.065 |
| CD4 [cels/mm3] [mean (SD)] | 680.79 (342.42 | 592.36 (343.71) | 0.154 |
| Viral load [logcopies/mL] [mean (SD)] | 2.34 (1.19) | 1.77 (0.66) | 0.037* |
| Mode of HIV acquisition | 0.588 | ||
| Parenteral drug use [n (%)] | 6 (12.8) | 4 (10.3) | |
| Heterosexual intercourse [n (%)] | 16 (34) | 12 (30.8) | |
| Homosexual intercourse [n (%)] | 19 (40.42) | 13 (33.3) | |
| Accidental [n (%)] | 0 (0%) | 1 (2.6%) | |
| Unknown [n (%)] | 6 (12.8%) | 9 (23.1%) | |
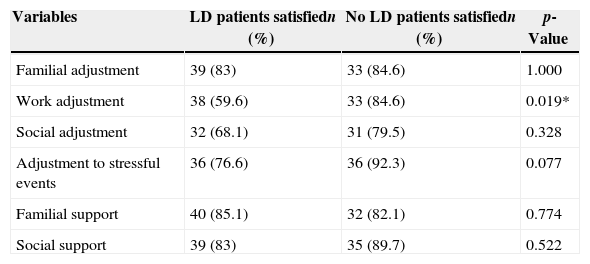
When studying psychosocial adjustment (detailed results in Table 2), LD patients referred worse work adjustment (p=0.019) as compared to non-LD patients.
Level of psychosocial adjustment of both groups [n=86; *p<0.05].
| Variables | LD patients satisfiedn (%) | No LD patients satisfiedn (%) | p-Value |
|---|---|---|---|
| Familial adjustment | 39 (83) | 33 (84.6) | 1.000 |
| Work adjustment | 38 (59.6) | 33 (84.6) | 0.019* |
| Social adjustment | 32 (68.1) | 31 (79.5) | 0.328 |
| Adjustment to stressful events | 36 (76.6) | 36 (92.3) | 0.077 |
| Familial support | 40 (85.1) | 32 (82.1) | 0.774 |
| Social support | 39 (83) | 35 (89.7) | 0.522 |
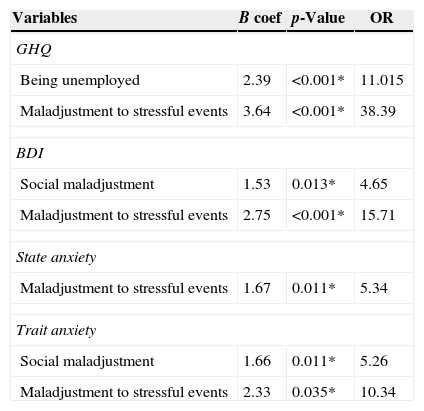
The mean score of the GHQ test for the total sample was 11.40 (SD 13.36); one-third of participants scored above the cut-off point. No statistically significant differences were obtained between groups. Multivariate analyses showed that being unemployed (p<0.001) and “maladjustment to stressful events” (p<0.001) were predictors of scores above the GHQ threshold criteria.
The mean score on the BDI for the total sample was 8.86 (SD 8.86). More than 30% of patients scored above the cut-off point, and no statistically significant differences were seen between groups either when comparing pathological scores (p=0.518) or mean scores (p=1). Multivariate analyses of BDI showed that “social maladjustment” (p=0.013) and “maladjustment to stressful events” (p<0.001) were predictors of psychopathological scores.
The mean score in the state anxiety scale was within the 38.37 percentile (SD 28.0). Pathological scores were observed in 27.9% of cases. LD patients showed higher statistically significant pathological scores (p=0.009) than non-LD patients. The logistic regression analyses showed that “maladjustment to stressful events” (p=0.011) was a predictor of a higher state anxiety score.
In the trait anxiety scale the mean score was in the 49.62 percentile and the SD was 33.14. Scores were above the cut-off point in 45.3% of patients. We observed no statistically significant differences between groups (p=0.131). When combining the variables in the logistic regression model, “social maladjustment” (p=0.011) and “maladjustment to stressful events” (p=0.035) were predictors of higher trait anxiety score.
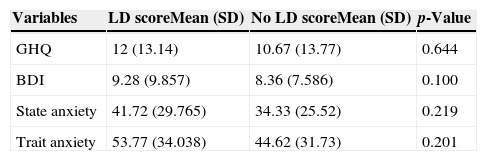
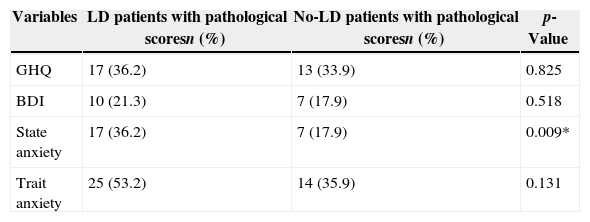
The mean scores on the questionnaires are summarized in Table 3. The percentage of pathological scores on each questionnaire is shown in Table 4. Finally, the predictors of psychopathology are described in detail in Table 5.
Detailed scores of the administered questionnaires [n=86; *p<0.05].
| Variables | LD scoreMean (SD) | No LD scoreMean (SD) | p-Value |
|---|---|---|---|
| GHQ | 12 (13.14) | 10.67 (13.77) | 0.644 |
| BDI | 9.28 (9.857) | 8.36 (7.586) | 0.100 |
| State anxiety | 41.72 (29.765) | 34.33 (25.52) | 0.219 |
| Trait anxiety | 53.77 (34.038) | 44.62 (31.73) | 0.201 |
Patients with pathological results of the administered questionnaires [n=86; *p<0.05].
| Variables | LD patients with pathological scoresn (%) | No-LD patients with pathological scoresn (%) | p-Value |
|---|---|---|---|
| GHQ | 17 (36.2) | 13 (33.9) | 0.825 |
| BDI | 10 (21.3) | 7 (17.9) | 0.518 |
| State anxiety | 17 (36.2) | 7 (17.9) | 0.009* |
| Trait anxiety | 25 (53.2) | 14 (35.9) | 0.131 |
Logistic regression analysis of psychopathological variables [n=86; *p<0.05].
| Variables | B coef | p-Value | OR |
|---|---|---|---|
| GHQ | |||
| Being unemployed | 2.39 | <0.001* | 11.015 |
| Maladjustment to stressful events | 3.64 | <0.001* | 38.39 |
| BDI | |||
| Social maladjustment | 1.53 | 0.013* | 4.65 |
| Maladjustment to stressful events | 2.75 | <0.001* | 15.71 |
| State anxiety | |||
| Maladjustment to stressful events | 1.67 | 0.011* | 5.34 |
| Trait anxiety | |||
| Social maladjustment | 1.66 | 0.011* | 5.26 |
| Maladjustment to stressful events | 2.33 | 0.035* | 10.34 |
In the current study we found that HIV-1 infected patients with LD had non-significantly higher pathological scores in the psychopathology questionnaires than non-LD patients. Scores for all tests were higher in both groups than scores reported in the literature for the general population.26,27
The GHQ scores for the total sample were pathological according to the aforementioned threshold criteria. These results are in line with many findings in the literature.13,14 When we compared the differences in psychopathology in LD and non-LD groups neither the mean scores nor the percentages of positive test scores were significantly different between groups. A more specific questionnaire would perhaps reveal psychopathological traits in LD patients that are not captured by the GHQ. Interestingly, patients with positive GHQ scores showed that “being unemployed” and “maladjustment to stressful events” were predictors of pathological scores in the GHQ in the multivariate analysis. This is in agreement with previous results from Lyketsos et al.18 and Hays et al.28 who found that being unemployed was a predictor of depression in HIV-1 infected patients.
Before HAART, the prevalence of depression in HIV-1 infected patients was reported to be much higher than in the general population, as stated by Lyketsos et al.18 and Judd et al.19 After 1996, HIV disease became treatable thanks to the availability of protease inhibitors, and the prevalence of depression decreased.20 Our results support these changes as the prevalence of depression was lower than that reported by Lyketsos et al.18 and Judd et al.19 We did not find any significant difference when comparing LD and non-LD patients, as stated by others.9 Authors such as Collins et al.3 and Power et al.7 suggest a relationship between LD and depression but these studies did not use validated instruments and depression was recorded only as a symptom. In our multivariate analysis “social maladjustment” and “maladjustment to stressful events” were positive predictors of pathological BDI scores, as reported previously by de Ridder et al.29 and Fehr et al.30
More than one-third of the total sample scored above the threshold criteria on both STAI scales, with the LD group showing higher values and higher percentages of pathological results than non-LD patients. Studies using the STAI to compare HIV-1 infected patients with the general population have found higher scores in the former but none of these reviews included LD patients.24 Power et al.7 performed a study with a small number of HIV-1 infected patients with LD using a semi-structured interview and found that fat redistribution was a predisposing factor for greater anxiety. Our analysis showed that “social maladjustment” and “maladjustment to stressful events” were predictors of pathological trait anxiety scores, while “being unemployed” and “maladjustment to stressful events” were predictors of pathological state anxiety scores. In our multiple regression model LD was not a predictor of trait anxiety or state anxiety. Although LD was not the most powerful predictor of anxiety, more detailed analyses in larger samples may reveal stronger associations since the whole HIV-1 sample had higher anxiety state scores than general population.
One of the limitations of this study is that it attempts to evaluate psychological aspects of a very complex syndrome. Our sample was small and we were unable to draw conclusions for subgroups of gender, age or LD severity. It should also be mentioned that the study was conducted in the outpatient service of a 600-bed tertiary university hospital in a cosmopolitan city where stigmatization concerning HIV infection has notably decreased in recent years. The sample is therefore not a true representation of the HIV community in our country as we did not include patients from smaller towns or rural areas where HIV infection and fat redistribution would perhaps have a deeper psychological impact due to higher stigmatization. A final limitation of this study is that we did not have a control group from the general population.
In conclusion, LD causes body disfigurement and has a profoundly negative impact on the psychological wellbeing and on the psychosocial adjustment of those affected. Although we did not find statistically significant differences between the two groups we think this is likely because variables such us LD severity, self-perception, gender and age were not included in the analysis. Studies with larger samples sizes are needed to better define the relationship between this very complex syndrome and its real psychological repercussions.
Conflict of interestThe authors declare no conflict of interest.