Natural products and their derivatives have been sources of search and research for new drugs for the treatment of neglected diseases. Naphthoquinones, a special group of quinones, are products of natural metabolites with a wide spectrum of biological activities and represent a group of interesting molecules for new therapeutic propositions. Among these compounds, lapachol stands out as a molecule from the heartwood of Tabebuia sp. whose structural changes resulted in compounds considered promising, such as epoxy-α-lapachone (ELAP). The biological activity of ELAP has been demonstrated, so far, for parasitic protozoa such as Leishmania spp., Trypanosoma cruzi and Plasmodium spp., species causing diseases needing new drug development and adequate health policy. This work gathers in vitro and in vivo studies on these parasites, as well as the toxicity profile, and the probable mechanisms of action elucidated until then. The potential of ELAP-based technology alternatives for a further drug is discussed here.
Neglected diseases (NDs) affect millions of people worldwide, especially in developing nations, which have little attention from their governments and pharmaceutical companies. The World Health Organization (WHO) has classified 20 of these diseases as neglected tropical diseases (NTDs), including several infections caused by viruses, bacteria, fungi, and protozoa.1,2
NDs mainly affect populations living in conditions of poverty in tropical and subtropical climates, with no adequate sanitation, precarious housing conditions and in contact with vectors.3 These diseases cause high morbidity and mortality rates, resulting in physical, economic and social impacts throughout life.1
Pharmaceutical companies have little interest in financing programs against these diseases, due to the low financial return they receive from these poor populations. However, programs and action plans for the control, elimination and eradication of these diseases have been developed, such as the WHO Sustainable Development Goals program, which aims to end tuberculosis, malaria and NTDs epidemics by 2030. An initiative to eliminate or eradicate 10 of these diseases by 2020 was presented in the WHO Roadmap on neglected tropical diseases and the 2012 London Declaration on Neglected Tropical Diseases. According to WHO, to achieve the objectives of these programs, public investments for the control of NTDs in the years from 2015 to 2020, excluding vector control, totaled an average of $750 million per year. To maintain progress from 2020 to 2030, an additional $460 million per year in investments are needed. Total investments, excluding donated drugs, for the 2015–2030 period total $34 billion.4
One focusof these control programs is the development of new therapeutic agents. However, the small advance in therapies and the high prevalence of NDs are still disproportionate. Despite the development of many compounds, few of them are directed to NDs.5 In this context, NDs caused by parasitic protozoa stand out in studies for the development of new therapeutic compounds. Although these diseases represent serious public health challenges, only a limited panel of drugs is commercially available for clinical applications.
The critical situation on Malaria, Chagas Disease and Leishmaniasis treatmentThe WHO warns of the increasing number of malaria cases in more than 80 countries and areas with continuous transmission of the disease.6 The treatment of malaria, a disease mainly caused by four human Plasmodium spp. (P. falciparum, P. vivax, P. malariae and P. ovale), has been progressively updated to the use of a combination of drugs. In this case, artemisinin-based combination therapy (ACT) is applied for infections caused by P. falciparum or by chloroquine-resistant strains of P. vivax, or by the use of chloroquine, supplemented with primaquine, for the treatment of chloroquine susceptible P. vivax infections.6
Chagas disease,caused by the protozoan Trypanosoma cruzi, is endemic in Latin American countries, affecting more than seven million people and causing more than 10,000 deaths per year.7 This infection causes irreversible and chronic damage to the heart, digestive system, and nervous system, with risk factors related to the low socioeconomic status of the affected population. Only two drugs have been approved for the treatment of Chagas disease during the 60s and 70s: nifurtimox, 5-nitrofuran, (Lampit™/Bayer) and benznidazole, a nitroimidazole (Lafepe and Abarax/Elea).8
Leishmaniasesare diseases caused by more than 20 species of protozoa belonging to the Leishmania genus.9,10 These parasites can affect the lining tissues, such as skin and mucous membranes (Cutaneous Leishmaniasis - CL), and some species can infect internal tissues and organs, such as liver, spleen, and bone marrow, as well as other organs such as kidney and lung, causing visceral leishmaniasis (VL).11,12 The disease is endemic in approximately 98 countries, especially in Latin America, East Africa and Southeast Asia, with 350 million people under risk of contracting the infection. The estimated incidence rate is 1.5 million new cases per year.9 Since 1940s until now, pentavalent antimonials (meglumine antimoniate and sodium stibogluconate) are considered the main drugs used to treat all clinical forms of leishmaniasis, especially in the New World.
These three groups of parasitic infections share a serious problem, the low availability of alternative treatments. Current treatment for these diseases is questionable and requires continuous monitoring. In addition, only a small portion of the infected population has access to these therapies. Futhermore, clinically approved drugs are expensive and toxic, need long-term administration and can induce the arise ofresistant strains.
In general, the treatment of these diseases showed little progress in the recent decades. For Chagas disease treatment, the use of benznidazole in children aged 2 to 12 years was approved by the US Food and Drug Administration (FDA) only in 2017.13 In 2014, the repositioning of miltefosine, initially developed for the treatment of breast cancer, was approved for the treatment of leishmaniasis, being currently the only oral medication available for this group of diseases.14 The use of tafenoquine for the radical cure of malaria caused by P. vivax was approved by the FDA in July 2018, being the first global drug for this indication in 60 years. In August 2018 its use for malaria prophylaxis was also approved.5,15 In 2020, FDA approved the intravenous use of artesunate in U.S. for the treatment of severe malaria followed by a full course of oral antimalarial treatments.16 In addition to these few important advances, the adverse effects and high toxicity that these drugs currently used present, make the need for the search for new therapies for these diseases even greater.
Insights into naphthoquinones as potential drugs to treat infection Plasmodium spp., Trypanosoma cruzi and Leishmania spp.Different strategies have been used for the search of new drugs for the treatment of these diseases, among them the research and use of natural products and their derivatives, mainly secondary metabolites.17 Natural products play an important role as source of compounds for new therapies. In this context, quinones are molecules of great interest in medicinal chemistry due to its spectrum of biological activity and chemical properties.18 Quinones are organic substances derived from natural aromatic metabolites found in several plant families, as well as in fungi, algae and bacteria. This group of compounds includes benzoquinones, anthraquinones and naphthoquinones.19
Among the natural naphthoquinones, lapachol (2-hydroxy-3-(3′-methyl-2-butenyl)−1,4-naphthoquinone) stands out. This molecule is known since 1882 and identified since then as a constituent of several plant species of the families Bignoniaceae, Verbenaceae and Proteaceae.20,21 The greatest occurrence is in the heartwood of the trunk of species of the genus Tabebuia, belonging to the family Bignoniaceae, popularly known as ipes.22,23 Lapachol has been tested in the past as an alternative in the treatment of solid tumors due to its anti-cancer properties.24 A range of biological effects of lapachol have been established which include analgesic, antiviral, antioxidant, antimicrobial, anti-inflammatory, fungicide and antiparasitic activities.25 Due to low toxicity, lapachol has become a good prototype for the development of synthetic naphthoquinones with interesting biological effects, often with equally low toxicity.25
The antiparasitic activity of lapachol and other naphthoquinones has already been demonstrated, as previously reviewed.26 In addition, other natural naphthoquinones are also notable for to their antiparasitic properties among them: α-lapachone, β-lapachone, lawsone, juglone and plumbagin.27 Several other studies have demonstrated the activity of these naphthoquinones against Leishmania spp.,28,29,30,31,32Plasmodium spp.33,34,35,36,37 and T. cruzi.38,39,40,41,42
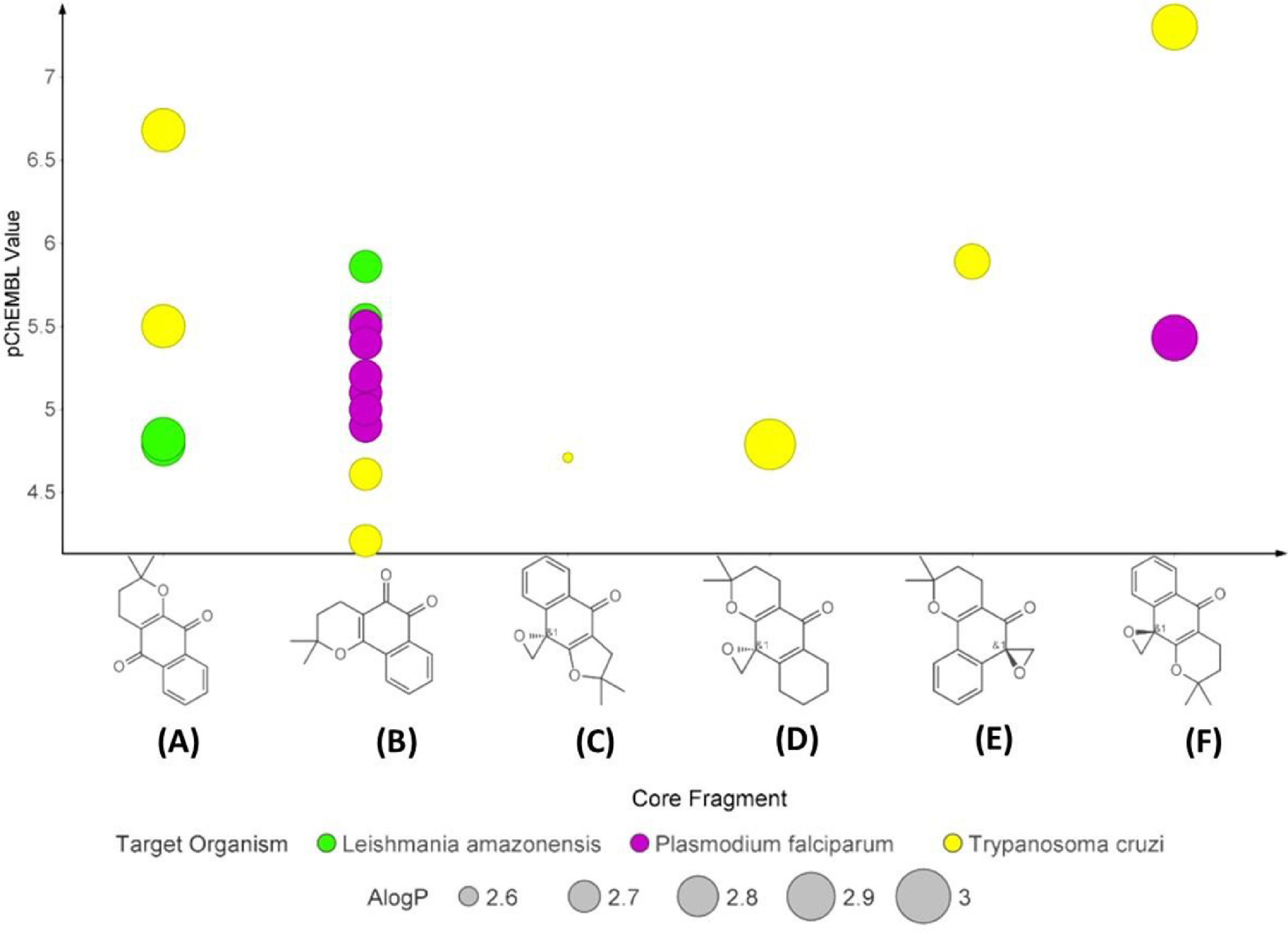
Supported by ChEMBL Database in vitro results against Leishmania spp., Plasmodium spp. and T. cruzi, six naphthoquinones were selected according to structural similarity, as shown in the plot on anti-parasitic activity (Fig. 1). Among these, two compounds are natural sources (α-lapachone and β-lapachone) and four are synthetic derivatives: 2,2-dimethylspiro[3H-benzo[f][1]benzofuran-9,2′-oxirane]−4-one, 2,2-dimethylspiro[3,4,6,7,8,9-hexahydrobenzo[g]chromene-10,2′-oxirane]−5-one, 2,2-dimethylspiro[3,4-dihydrobenzo[h]chromene-6,2′-oxirane]−5-one and epoxy-α-lapachone. Fig. 2 shows that α-lapachone, β-lapachone and epoxy-α-lapachone have been the subject of careful studies against these parasites and the number of in vitro studies on the determination of IC50 values stands out.
Naphthoquinone anti-parasitic activity plot based on experimental assays. The compounds α-lapachone (A), β-lapachone (B), 2,2-dimethylspiro[3H-benzo[f][1]benzofuran-9,2′-oxirane]-4-one (C), 2,2-dimethylspiro[3,4,6,7,8,9-hexahydrobenzo[g]chromene-10,2′-oxirane]-5-one (D), 2,2-dimethylspiro[3,4-dihydrobenzo[h]chromene-6,2′-oxirane]-5-one (E) and epoxy-α -lapachone (F) were selected according to structural similarity (>50%) using the ChEMBL Database (https://www.ebi.ac.uk/chembl/). This analysis shows the activities of these molecules against to Plasmodium spp. (purple circle), Trypanosoma cruzi (yellow circle), and Leishmania spp. (green circle) based on IC50 values normalized (pChEMBL value). The circle sizes show a predict values of permeability (ALogP) of these compounds.
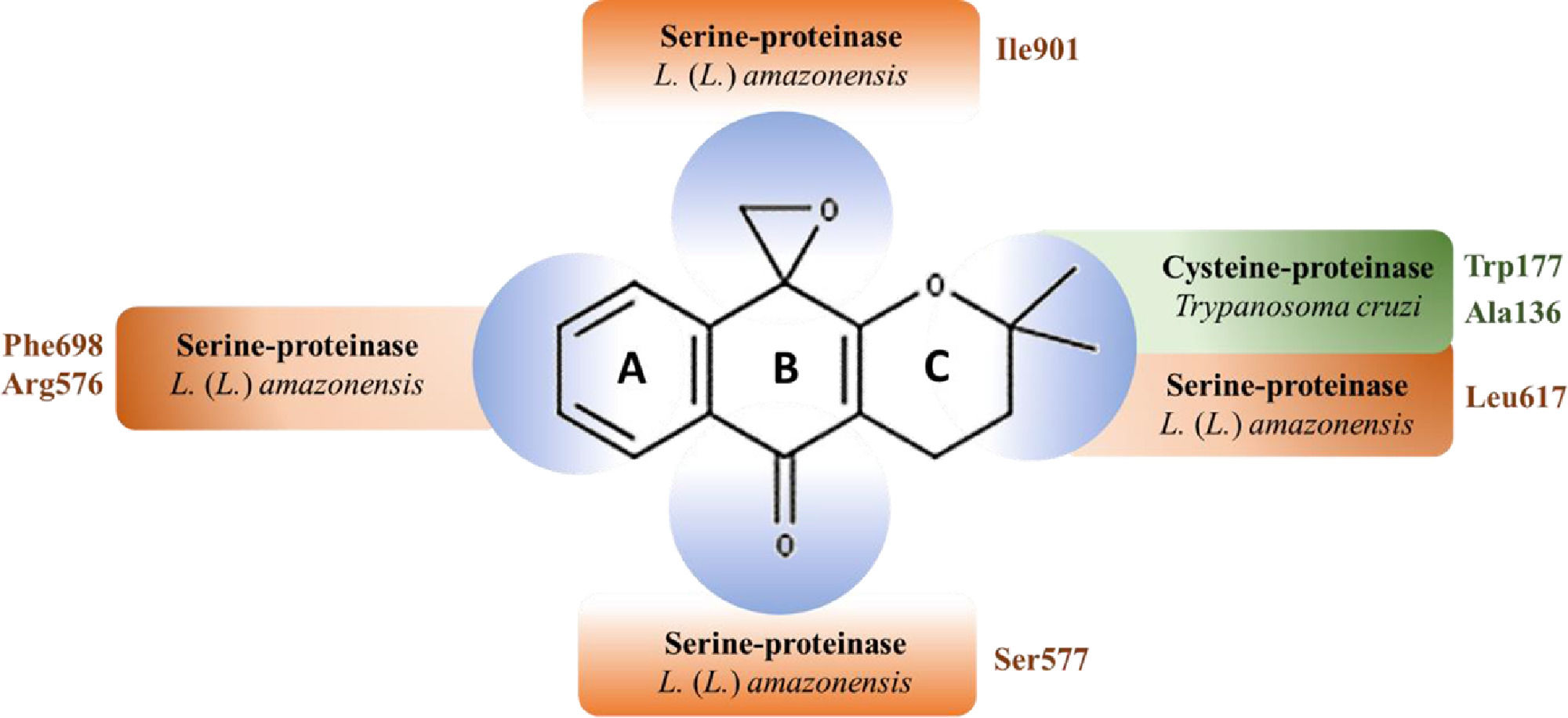
Predicted epoxy-α-lapachone regions interactions with amino acid residues of the active site of trypanosomatid proteases. Epoxy-α-lapachone (2,3-Dihydro-3,3-dimethylspiro[1H-4-oxanthracene-5,2′-oxiran]−10(5H)-one) is formed by an aromatic ring (A), a central six-membered ring (B) and a ring with two methyl groups (C). The chiral center being the C12 atom, which is also part of the three-membered epoxide ring.44 CID: 12,000,280; molecular formula:C16H16O3 and molecular mass: 256.3 g/mol. Epoxy-α-lapachone regions interactions with amino acid residues of cysteine-proteinase (green) of Trypanosoma cruzi48 and serine-proteinase (orange) of Leishmania (Leishmania) amazonensis.50
The toxicity of β-lapachone has led to the study of synthetic and semi-synthetic derivatives which may avoid this disadvantage.43 This review examines epoxy-α-lapachone (ELAP) in which an epoxide ring is introduced into the quinoid center of α-lapachone.44
Epoxy-α-lapachoneSynthesis and chemical characterizationEpoxy-α-lapachone (2,2-Dimethyl-3,4-dihydro-spiro[2H-naphtho[2,3-b]pyran-10,2′-oxirane]−5(10H)-one), ELAP, molecular weight 256.3 g/mol and molecular formula C16H16O3, synthesized by the reaction of α-lapachone with an ethereal solution of diazomethane, the first spiro-oxirane derived from a p-quinone to be reported.44 This mechanistic consideration resulted in obtaining ELAP that maintained the parasiticidal activity of its precursor.45
Antiparasitic activityELAP activity was assessed against three protozoan species: Trypanosoma cruzi, Leishmania spp. and Plasmodium falciparum, causative agents of Chagas disease, leishmaniasis and malaria, respectively. The summary of the main results of in vitro ELAP assays is shown in Table 1.
Main in vitro biological activities of epoxy-α-lapachone.
| Species | Evolutive form | Biological Activity | References |
|---|---|---|---|
| Tripanosoma cruzi | Epimastigote (strain Dm28c) | LD50 < 3.1 µM - 72 h | 46 |
| IC50 = 1.3 µM - 72 h | 47 | ||
| Growth inhibition (100%) - 72 h | 48 | ||
| Epimastigote (strain Y) | IC50 = 0.05 µM - 72 h | 39 | |
| Tripomastigote (strain Y) | Growth inhibition (97%) - 72h | 49 | |
| Tripomastigote (strain Colombiana) | Growth inhibition (84%) - 72 h | ||
| Intracellular amastigote (strain Y) | Human macrophages:Growth inhibition (85.6%) – 72 hVERO Cell:Growth inhibition (96.4%) – 72 h | ||
| Intracellular amastigote (strain Colombian) | Human macrophages:Growth inhibition (71.9%) – 72 hVERO Cell:Growth inhibition (95.0%) – 72 h | ||
| Leishmania (Leishmania) amazonensis (strain MHOM/BR/73/LTB0016)Leishmania (Viannia) braziliensis (strain MCAN/BR/1998/R619) | Promastigote | IC50:L. (L.) braziliensis: 37.0 ± 0.4 µM – 24 hL. (L.) amazonensis: 37.0 ± 0.4 µM – 24 h | 31 |
| Intracellular amastigote | Human macrophages - IE:L. (L.) braziliensis: 21.0 ± 2L. (L.) amazonensis: 6.0 ± 0.8 | ||
| Plasmodium falciparum | Erythrocytic form(strain 3D7) | IC50 = 3.71 μM - 48 h | 37 |
The effect of ELAP on the viability of intracellular epimastigotes, trypomastigotes and amastigotes of T. cruzi was assessed. ELAP showed high activity against epimastigote forms (Dm28c strain), eliminating all parasites within 72 h (DL50 < 3.1 μM).46 The compound showed the highest activity against this parasite form (IC50 = 1.3 μM) in the series of tested oxiranes.47 ELAP affected the viability of 100% of epimastigote forms when tested in concentrations of 3.1 μM, 12.5 μM and 50 μM.48
ELAP showed a lower IC50 value (IC50 = 0.05 μM) than epoxymethyl-lawsone (IC50 = 1.13 μM), an oxirane derived from 2-hydroxy-1,4-naphthoquinone (lawsone) after 72 h of exposure. Both molecules showed lower IC50 values than benznidazole (IC50 = 11.5 μM), the drug of choice for the treatment of Chagas disease.39
Their effects on trypomastigote and intracellular amastigote forms of the Y and Colombian strains of T. cruzi, known for their different infectious profiles, were assessed. This study demonstrated that 75 μM of the compound has affected the viability of both strains: 97% of strain Y and 84% of Colombian strain. ELAP activity against intracellular amastigotes inVERO cells infected was higher (96.4% for strain Y, and 95.0% for Colombian strain) than to the human macrophages infected (85.6% strain Y and 71.9% Colombian strain). Interestingly, results of these assays suggest a preferential in vitro order of ELAP activity against the three T. cruzi forms: epimastigotes > trypomastigotes > intracellular amastigotes.49
Leishmanicidal activity of ELAP was evaluated against L. (V.) braziliensis and L. (L.) amazonensis, two of the main causative species of CL in the New World. Promastigote assays showed that ELAP was able to significantly decrease the number of promastigotes after 24 h of exposure, compared to the control, indicating a similar IC50 value (37.0 ± 0.4 μM) for both parasite species. A higher effect of ELAP against these forms in 48 h of exposure indicates that the activity is time- and dose-dependent.31 The effect of ELAP against intracellular amastigote forms was also investigated and a reduction in the endocytic index values for L. (V.) braziliensis (491.1 ± 40 to 21.0 ± 2) and for L. (L.) amazonensis (290.0 ± 30 to 6.0 ± 0.8) was observed, demonstrating the ability of the compound to cross the macrophage cell membrane and affect the parasite.31 In addition, it was noted that ELAP is able to induce changes in the mitochondrial membrane potential of the parasite.50
A study using BALB/c mice infected with L. (L.) amazonensis demonstrated that ELAP (0.44 mM) administered subcutaneously in the dorsal region was able to reduce the size of paw lesions to 18% six weeks after treatment compared to untreated animals group (30.8 ± 2.6 mm3) and animals treated with meglumine antimoniate (MA), (28.3 ± 1.5 mm3).50
ELAP and 17 other compounds derived from quinones were also tested on the 3D7 strain of P. falciparum present in erythrocytes (100 to 0.14 μg / mL). The results indicated that ELAP is the most active compound against this parasite, presenting the lowest IC50 value (3.71 μM), followed by another oxirane compound derived from a tetrachlorobenzoquinone whose IC50 value was 3.95 μM.37
Combination therapyCombination therapy has been the basis for the treatment of several infectious diseases, such as malaria, tuberculosis, HIV/AIDS, and its use has gradually been expanded to NTDs, such as leishmaniasis and Chagas disease.51,52,53,54 This approach has several advantages, such as increased efficacy, reduced dose and treatment time, lower incidence of adverse effects, better patient adherence to treatment, and improved cost-effectiveness. In addition, this approach reduces the possibility of resistant parasite selection.51,54,55
The combination of naphthoquinones and their derivatives with other compounds have been tested in other studies as an alternative to the conventional treatment of leishmaniasis. Among them, atovaquone presented leishmanicidal effects in vivo when combined with pentavalent antimonials on L. (L.) donovani as complement to the conventional treatment of VL.56 Although L. (L.) infantum promastigotes showed resistance to atovaquone when subjected to strain selection (after the fifth pressure), further studies may confirm that the use of atovaquone in combination with approved drugs may still be an alternative for the treatment.57 Plumbagin, a naphthoquinone obtained from the roots of Plumbago capensis, was tested in a double combination with acriflavine, saponin or trifluralin, for the treatment of BALB/c mice infected by L. (L.) major. Combination therapy resulted in the total elimination of parasites in the lesions and significantly reduced the parasitic burden on the liver and spleen, compared to monotherapy and untreated controls.58
Interestingly, ELAP activity was more effective when the treatment was performed in combination with MA compared to monotherapy. The combination of MA/ELAP (3:1) caused a 98% reduction in the in vitro endocytic index of murine peritoneal macrophages infected with L. (L.) amazonensis. The treatment of BALB/c mice infected with the same species resulted in a reduction of 62% of the size lesion of the paw, and decrease of 97% in parasitic load in the paw.59 These results indicate that the combination of these compounds with pentavalent antimonials may represent a promising approach to the management of CL treatment.
ToxicityThe added value of toxicity tests during research and development of new drugs, represents a major contribution to the understanding of the dose-response relationship as well as to the extrapolation of data obtained in research with animal models for humans.60 In this way, a set of in vitro and in vivo tests has been conducted to establish the safety range for research on ELAP as a potential drug.
The in vitro cytotoxicity of ELAP was assayed on VERO cells (ATCC, CRL-1586), a fibroblast cell line in the kidney of the African green monkey (Cercopithecus aethiops). ELAP was shown to be the least cytotoxic compound (CD50> 50 μM) compared to other tested naphthoquinone derivatives.46 Lower cytotoxicity and high selectivity (CC50/IC50) were found for ELAP (CC50> 50 μM and IC50 = 1.3 μM) when compared to β-lapachone (CC50 <3.1 μM and IC50 = 0.9 μM).47 ELAP presented no toxicity to bone marrow-derived macrophage lineage, and also to VERO cells as noted in previous studies.48,49
ELAP was tested on a human fibroblast cell line (MRC-5) and human macrophages. The assays with MRC-5, for 48 h of interaction in the concentrations of 1.56 μg/mL to 100 μg/mL, presented a low IC50 value on this strain (IC50> 100 μM) and a significant selectivity index (IS> 27), when compared to the other compounds tested.37 Tests over human macrophages from peripheral blood for 24 and 48 h, showed no effects on the viability of this cell type at tested concentrations (25 μM and 75 μM).31
The effects of ELAP (1.9 μM/kg/day) in the organs of healthy BALB/c mice were also investigated, which allowed to evaluate the nature of the damage that these compounds may cause at high doses. Heart tissue was most affected by ELAP with intense necrosis, degeneration of cardiac fibers and mononuclear infiltrates.61
Mechanism of action on the protozoan parasitesThe pharmacological activity of a drug is directly related to its chemical structure, which guides the interaction with the parasite's binding site. It is well known that an important structural feature for the biological activity of α-lapachone is the presence of the C ring as well as the strong influence of the redox center.47 Through structural changes in the α-lapachone molecule, several compounds were synthesized, including ELAP, by the introduction of the oxirane ring in the quinonoid center.47
To elucidate the mechanism of action of ELAP, as well as other derivatives, previous data about the activities of its precursors are important. Differently from other quinones, whose mechanism of action is related to the formation of ROS from the redox cycle of the ortho- and para-quinonoid centers, the activity of α-lapachone and its derivatives has not been shown to be involved in the ROS production, especially in the T. cruzi.46,62,63,64,65 It has also been proposed that the trypanocidal activity of these compounds would be due to another mechanism, possibly related to the presence of the oxirane ring of ELAP, since this modification resulted in increased cytotoxic effect on the parasite.46
A possible mechanism for ELAP action is an inhibition of serine proteases of the parasite, observed in epimastigote forms of T. cruzi and in both promastigotes and amastigotes of L. (L.) amazonensis.48,50 Furthermore, in silico studies indicate that ELAP is able to bind oligopeptidase B (OPB) from L. (L.) amazonensis.50
Although the mode of action of ELAP on biochemical pathways of P. falciparum is still unknown, it is possible to hypothesize such actions based on the findings in T. cruzi, L. (L.) amazonensis and L. (V.) braziliensis. There is evidence that ELAP acts as a serine protease inhibitor of P. falciparum,37 but nothing has been described about the effect of the compound on the multiplication or any other event in the parasite's biological cycle. However, it is plausible that its action interferes with several points in the parasite's physiology, since proteolytic activity, including the numerous isoforms and homologues of serine proteases, is essential for the survival of organisms.66
In addition to these biological actions described for ELAP, it is also possible to conjecture its role in the modulation of cells or components of the immune system, as discussed based on the reactive potential of its molecular structure.59 Thus, it is possible that ELAP acts directly over the parasite cell or indirectly by the modulation of mediator's expression of the immune response, as proposed for α-lapachone and β-lapachone. For both isomers, properties of blocking the expression of pro-inflammatory cytokines such as interleukin IL-1b, IL-6 and tumor necrosis factor have been described.67
A recent in silico study demonstrated the potential of ELAP to act on different enzymes from Leishmania spp. based on previous results in the literature related to the activity of other naphthoquinones in different parasites: β-lapachone in Coccidioides posadasii; 2-phenoxy-1,4-naphthoquinone in T. brucei; Buparvaquone in L. (L.) mexicana.68 Docking results showed that ELAP is able to form stable complexes with favorable binding energy with key enzymes of the metabolic pathway such as glycolysis (glyceraldehyde 3-phosphate dehydrogenase: −8.5 kcal/mol to −8.3 kcal/ mol); electron transport chain (Cytochrome C: −10.0 kcal/mol to −9.0 kcal/mol); and lipid metabolism (lanosterol C-14 demethylase: −8.4 kcal/mol to −8.2 kcal/mol) of Leishmania spp.68
Conclusion and remarksMore effective drugs with low toxicity for the treatment of NDs are needed, which has encouraged studies with natural products and their derivatives as potential sources. Thereby, the results presented in this review indicate safety and efficacy of ELAP as a promising drug for use in the treatment of different parasitic diseases, such as malaria, Chagas disease and leishmaniasis. The accumulated knowledge in the past 19 years about the successful tests with ELAP compound in preclinical trials for the treatment of these NTDs motivated the present review study, highlighting a potential multi-target drug for these parasites, based on its notable chemical property and the ability to act as an oxidizing or dehydrogenating agent.
To the best of our knowledge, in vitro assays and studies with infection models, aiming to establish the efficacy and understanding of the mode of action is more advanced for tegumentary leishmaniasis compared to those related to malaria and Chagas disease. Available expertise on the Leishmania spp. infection model shows evidence that supports the potential of ELAP as a multi-directed compound capable of interrupting the network interactions of important parasite enzymes, representing a promising approach for the treatment of these parasitic diseases. These data certainly point to the possibility of combination therapy of ELAP with MA for the treatment of CL,59 since different chemical structures and mechanisms of action of both drugs may interfere on multiple physiological targets, obtaining a synergistic or additive effect on the parasites.68
Furthermore, the combination of ELAP and pentavalent antimonials might represent a therapeutic line with a better balance between effectiveness and toxicity, preventing or minimizing some known adverse effects of current therapy. In addition, this approach can avoid the appearance of refractory strains. In this context, there is the possibility to study the use of ELAP combined with traditional therapies for other NDs.
Aiming to improve the effects on parasites and reduce the incidence of adverse effects, there is also the alternative to incorporate ELAP in drug delivery systems based on micro and nanotechnology. This approach can increase stability and bioavailability as well as promote a more efficient release of the compound.69,70,71,72
Data gathered here show evidence that studies with Leishmania spp. provide the necessary information for conducting a high technology readiness level (TRL)/ manufacturing readiness level (MRL),73 sinceproofs-of-concept have have already been completed. Thus, the set of results gathered available to the scientific community indicate ELAP as a viable product worthy of development as a drug.
This study was financed in part by the Conselho Nacional de Desenvolvimento Científico e Tecnológico - Brasil (CNPq: 301744/2019-0 and 152616/2022-5), Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro - Brasil (FAPERJ: E-26/010.000983/2019; E-26/200.799/2021; E-26/201.654/2021) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil (CAPES) - Finance Code 001. Carlos R. Alves is research fellows at CNPq institution.



![Naphthoquinone anti-parasitic activity plot based on experimental assays. The compounds α-lapachone (A), β-lapachone (B), 2,2-dimethylspiro[3H-benzo[f][1]benzofuran-9,2′-oxirane]-4-one (C), 2,2-dimethylspiro[3,4,6,7,8,9-hexahydrobenzo[g]chromene-10,2′-oxirane]-5-one (D), 2,2-dimethylspiro[3,4-dihydrobenzo[h]chromene-6,2′-oxirane]-5-one (E) and epoxy-α -lapachone (F) were selected according to structural similarity (>50%) using the ChEMBL Database (https://www.ebi.ac.uk/chembl/). This analysis shows the activities of these molecules against to Plasmodium spp. (purple circle), Trypanosoma cruzi (yellow circle), and Leishmania spp. (green circle) based on IC50 values normalized (pChEMBL value). The circle sizes show a predict values of permeability (ALogP) of these compounds. Naphthoquinone anti-parasitic activity plot based on experimental assays. The compounds α-lapachone (A), β-lapachone (B), 2,2-dimethylspiro[3H-benzo[f][1]benzofuran-9,2′-oxirane]-4-one (C), 2,2-dimethylspiro[3,4,6,7,8,9-hexahydrobenzo[g]chromene-10,2′-oxirane]-5-one (D), 2,2-dimethylspiro[3,4-dihydrobenzo[h]chromene-6,2′-oxirane]-5-one (E) and epoxy-α -lapachone (F) were selected according to structural similarity (>50%) using the ChEMBL Database (https://www.ebi.ac.uk/chembl/). This analysis shows the activities of these molecules against to Plasmodium spp. (purple circle), Trypanosoma cruzi (yellow circle), and Leishmania spp. (green circle) based on IC50 values normalized (pChEMBL value). The circle sizes show a predict values of permeability (ALogP) of these compounds.](https://static.elsevier.es/multimedia/14138670/0000002700000002/v3_202304071918/S1413867023000041/v3_202304071918/en/main.assets/thumbnail/gr1.jpeg?xkr=ue/ImdikoIMrsJoerZ+w95uaF0+42b+pWE4hY44gaZY=)
![Predicted epoxy-α-lapachone regions interactions with amino acid residues of the active site of trypanosomatid proteases. Epoxy-α-lapachone (2,3-Dihydro-3,3-dimethylspiro[1H-4-oxanthracene-5,2′-oxiran]−10(5H)-one) is formed by an aromatic ring (A), a central six-membered ring (B) and a ring with two methyl groups (C). The chiral center being the C12 atom, which is also part of the three-membered epoxide ring.44 CID: 12,000,280; molecular formula:C16H16O3 and molecular mass: 256.3 g/mol. Epoxy-α-lapachone regions interactions with amino acid residues of cysteine-proteinase (green) of Trypanosoma cruzi48 and serine-proteinase (orange) of Leishmania (Leishmania) amazonensis.50 Predicted epoxy-α-lapachone regions interactions with amino acid residues of the active site of trypanosomatid proteases. Epoxy-α-lapachone (2,3-Dihydro-3,3-dimethylspiro[1H-4-oxanthracene-5,2′-oxiran]−10(5H)-one) is formed by an aromatic ring (A), a central six-membered ring (B) and a ring with two methyl groups (C). The chiral center being the C12 atom, which is also part of the three-membered epoxide ring.44 CID: 12,000,280; molecular formula:C16H16O3 and molecular mass: 256.3 g/mol. Epoxy-α-lapachone regions interactions with amino acid residues of cysteine-proteinase (green) of Trypanosoma cruzi48 and serine-proteinase (orange) of Leishmania (Leishmania) amazonensis.50](https://static.elsevier.es/multimedia/14138670/0000002700000002/v3_202304071918/S1413867023000041/v3_202304071918/en/main.assets/thumbnail/gr2.jpeg?xkr=ue/ImdikoIMrsJoerZ+w95uaF0+42b+pWE4hY44gaZY=)


